Introduction
Recently the basis of utilizing antiretroviral treatment for South African citizens suffering from AIDS has been use of highly active antiretroviral treatment (HAART), which consists of two nucleoside formulations along with either a non nucleoside formulation or a protease inhibitor provided concurrently to persons suffering from Aids on a continuing mainstay.
HAART is considered to be more effective to reduce HIV progression and morbidity in relation to other analogues like nucleoside reverse transcriptase inhibitor (NTRI) [1]. However, constantly mounting pressure on clinical service monetary allocations makes it necessary that clinical service technology shows not only wellbeing and effectiveness but in addition cost efficacy.
Financial analyses bring about details regarding cost effectiveness through contrasting the cost and gains of a single medical service project to the cost and/or advantageous of an agenda that it is at the end targeting to substitute. Although highly active antiretroviral treatment is currently popular therapy for people suffering from AIDS in South Africa, insufficient proof was documented regarding its cost efficacy before its extensive utilization.
Furthermore, as it has of late been proposed that the guideline for financial analysis should be dynamic and needs to proceed prior to technologies has become reference healthcare policy [2-5] instead of a single event, the study aims to examine the cost efficacy of HAART in South Africa.
Methods
Study modeling
A Markov modeling comprising 38 twelve-month durations was utilized in simulating the progression regarding HIV scourge and in estimating the cost, effect and cost efficacy [4, 6] of highly active antiretroviral treatment alternative [7]. The design of the Markov modeling was based on frameworks earlier utilized in approximating the cost efficacy of HAART [8, 9].
Markov states were developed as per the CD4 numbers, which offer evidence upon which cost efficacy of AIDS therapy is modeled. Cost efficacy was examined through dividing the cost variance with the variance that occurs in health results, that is, quality adjusted life year (QALY) or life year (LY) added between the HAART and No-HAART alternatives in producing incremental cost efficacy ratios (ICERs) [5].
Probabilistic simulations (Monte-Carlo) evaluation was developed based on an imaginary sample of 2000 AIDS patients (1000 in HAART cohort and 1000 in No-HAART cohort) who were aged 18 when they entered the Markov modeling, thus enabling investigation on the impacts of indecision regarding the various variables.
The simulations were carried out numerous times to enable representation of a number of various medical events during evaluations. The reference cost of No-HAART treatment, time of HAART therapy impact and the consumption weightings were stated to be mainly “possible” values, in combination to lower-and-upper boundaries, instead of one-point approximates, in a quest of describing more practical events [3, 10].
A more potential value was set equivalent to documented average value while lower and upper boundaries were set at 0.95 confidence levels. The Markov modeling comprised 4 health conditions: 200<=CD4>350 cell/micro liter, 50<CD4<200 cell/micro litre, AIDS and Death. Patients entered the Markov model at the health condition 200<=CD4>350 cell/micro liter. After each cycle, patients’ HIV state either remained unchanged, advanced or reversed.
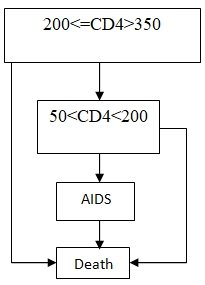
Figure 1: Health conditions (see rectangles) and transiting probability (see arrows) comprised in Markov modeling (arrow indicates direction of possible transition)
Therapy impacts (transiting probability)
Overall effect of therapy on HIV progressions were evaluated utilizing examinable data from South Africa. Information on ART naive patients who commenced using HAART and who in addition had 200<=CD4>350 cell/micro liter formed part of the cohort. For patients getting HAART treatment, HIV progressions were examined utilizing data from 1000 patients who enrolled for ART therapy over the research duration.
HAART effect on HIV progressions were examined utilizing details on 1000 patients who commenced full antiretroviral treatment. Follow-ups for such patients were expurgated during their final visit and/or death, and comprised information prior to treatment, as therapy adjustments at this period would have no impact on progression probability. The uncertainty to die was accounted not only for the concerned age-adjusted death degree in the sample but also for the threat of receiving AIDS [5].
For the sake of this study, a patient in the two cohorts who passed on during treatment duration was deemed to have passed on because of an AIDS-based circumstance. However, this can over approximate the death cases linked to HAART, since a larger percentage of death cases are expected to be non-AIDS-based in this cohort [9, 11].
Antiretroviral unresponsive people suffering from AIDS are regarded to be having an enhanced first reaction to therapy in relation to people who have earlier been introduced to antiretroviral [2, 3, 9]. Thus, the model comprised transiting probabilities from one health state to another (see table 1).
Because individual follow ups were uncensored when a patient progression along the Markov cycles, the modeling absolutely integrates succeeding therapy malfunction and adjustments during treatment. The first and succeeding pair of transiting likelihoods in each therapy was computed by classifying patients as per their initial health status, 12 months and during 24 months after commencing therapy.
A patient who passed on during the 12 months was added to the death state while a patient who had opportunistic disease or whose final HIV based condition was over ½ a year prior to the expected time, were grouped as per their CD4 amount during the event (200<=CD4>350 cell/micro litre, 50<CD4<200 cell/micro litre).
The CD4 counts during each period were approximated utilizing linear regression between the prevailing counts and counts after the specified time period. In case no CD4 values were found after this period, the CD4 counts were approximated to be the number prior to this period, as long as the value was determined during the first quarter of that period [12].
Finally, since the period of the impact of HAART is greatly not known, in the basic evaluation it was presumed that extra medical effects of HAART were assumed to be for 60 months with a least and optimum period of 24 and 96 months, in that order. However, the extra cost of HAART in relation to No-HAART treatment was presumed to be separate from medical impact and to prolong up to either the modeling stopped or a person passed on, whichever came early, so as to create conventional estimates of cost efficacy [13, 14].
Table 1: Yearly transiting probability based on a group of persons suffering from AIDS who were listed for therapy in South Africa
Transition probability
Changeover probability based on Markov modeling is needed in specifying all necessary transitions of Markov conditions (see arrows in fig.1). For the HAART modeling, movement probability as well as 0.95 confidence interval (CI) is approximated based on Kaplan result maximum approximations of survival representing 1000 participants under antiretroviral for the initial 4 years of the study [7, 8].
Changeover probability for the No-HAART modeling was gotten from a domestic ordinary history group encompassing 1000 ART unresponsive participants. The probability of transiting from 200<=CD4<350 to 50<CD4<200 is 0.21, probability of transiting from 50<CD4<200 to AIDS is 0.45, probability of transiting from AIDS to death is 0.4, probability of transiting from 200 <=CD4<350 to Death is 0.2, and that of transiting from 50 <CD4<200 to Death is 0.25.
Treatment cost
The cost to treat people suffering from HIV with HAART treatment depended on approximates gotten from the South African Potential Evaluating System-AIDS Health Financial research [14]. The research approximated the HIV treatment requirements that were needed by more than 13 000 people suffering from AIDS in 9 hospitals in South Africa, comprising the resources attached to related diseases. Resource approximates were the associated with basic individual costs gotten from an earlier research [12].
This cost was in addition combined with approximates of the social service cost linked to various phases of HIV [15], implying that such an evaluation was carried out based on South African public monetary view. The costs to treat people using HAART were approximated as the cumulative costs to treat people with HAART plus the costs to treat HIV related complications. Discounted per case life-time cost were 2,232,614 for No-HAART against 8,131,018 for HAART.
The discounted incremental cost (ICER) per QALY added was 10,533. The ICER value was less than per life year added and fairly greater after discounting costs at 3%. No additional cost was added to this assessment. In the basic evaluation, cost was discounted at 3% in a year but medication impact remained undiscounted, as per the current South African policies, apart from the sensitivity assessment [6, 7]. All documented expenditures were based on 2008 data [12].
Table 2: Total costs of HAART treatment in 2008 (US dollars) and utility weightings based on HIV condition [12].
Quality of life
Currently, only a few utility weightings are available to people suffering from AIDS. The most appropriate information was generated from a cohort comprising 249 Canadians suffering from HIV utilizing the health utility indexes mark II [12].
However, except for the fact that such information was generated for Canadian individuals, it was only evaluated utilizing multivariate modeling, implying that absence of variations in documented weightings between these two cohorts could be described using elements like before therapy background, period of the last HIV associated situation, threat class or opportunistic diseases.
The Mark II comprises of a health condition categorization method and a corresponding pair of utility weightings [12, 13]. The categorization method has seven measures, consisting of mobility, pain and healthcare. The utility weighting was computed through requesting 1000 Canadians to respond to a visual-analogue scale and through scoring the responses using standardized gable value.
Sensitivity evaluation
Basically, the greatness of the ICERs measured against their compound variables, and therefore the certainty level that may be associated with the basic cost efficacy approximate, can be examined utilizing sensitivity evaluation [4, 15]. In this study, 1-way sensitivity evaluations were carried out on different elements, comprising the extra costs associated with HAART in order to emphasize their significance to determine cost efficacy.
This was attained through varying the number associated with one element (for example cost of HAART) while leaving all other numbers in the modeling process constant, and through analyzing the succeeding variation in ICER value. Elasticity, which measures the effect of adjusting various parameters on the ICERs, was in addition computed for some elements like the ICER’s % change against a % change in the variable.
Overall, a higher elasticity means an enhanced importance of the variable to determine cost efficacy. Negative elasticity is linked to a decrease in the ICER, that is, an increase in cost efficacy, while positive elasticity indicates that the ICER value increased, implying a reduction in cost efficacy.
Analysis/Results
In this study evaluation, the cumulative cost, life years and QALYs for people who did not use HAART for 38 years was 2232614, 90 and 1964, in that order (see table 3), and for people treated with HAART, the cost, life years and QALYs linked to therapy was 8131018, 115 and 2524 respectively, implying that the cost for treating individuals and impacts were boosted by approximately 36.3 per cent and 22.1 per cent, in that order, generating an ICER value of 238543 for one year added.
Approximately 24 % of the cost of highly active antiretroviral treatment was due to the costs to provide HIV drugs. Adjusting life years in the health conditions resulted to an ICER value of 10533 in each QALY added (Table 3).
Also the cost per case amounted to $138 for participants under HAART and $250 for patients not under HAART. The mean cost of each patient case amounted to $267. 71% of patients led to a mean weighted cost amounting to $162 per case. The cost of each patient treated amounted to $622.
The percentage for each case cost associated with treatment varied between 4-34% for patients under HAART. The total yearly cost for HAART including cost of drugs amounted to $438 in a year whereas the highly active antiretroviral therapy cost totaled to $162 in a year. The second phase was twice the cost of first phase, amounting to $952 in a year. The cost of checking CD4 and drug costs amounted to approximately $25 in each quarter.
The cost in each Markov state has been computed through multiplying health service use and per case costs. HAART cost was greatest for patients bearing 50<CD4<200 during the initial 12 weeks under HAART, amounting to $548 not including cost for dead patients.
Cost remained steady, but shifted to more than $340 in every year period after which second-phase medication was included. No-HAART cost in the 50<CD4<200 and 200<=CD4>350 classes amounted to $250 (undiscounted) and to $223 with discounts using an overall discount factor of 22.
Table 3: Cost, impact and cost efficacy of HAART in relation to No-HAART
The outcomes indicated that the ICER values were slightly elastic to a one percent increase of the per year probabilities of HIV advancement and fairly elastic upon increasing the cost of HAART (with a sensitivity of 0.68). However, additional evaluation indicated that the ICER values were highly elastic to increasing rate of discounted cost (with a sensitivity of -7.1) and also sensitive to a discount of treating impacts per year at 1% (with a sensitivity of 10.7).
The ICERs were in addition highly elastic to the presumption about the time and costs provided that HAART prolonged in having an incremental impact, the ICERs declined to about 238543 in each life year added. Furthermore, when the probable time periods of the effectiveness of HAART were extended to six years from five, the ICERs declined to about 229070 in each life year added.
Discussion
This study aims at approximating the cost efficacy of complete AIDS healthcare comprising HAART based on principal costs, use, Life years and life-adjusted quality years data derived from an extensive group in an industrializing nation setup.
This research has initiated some improvements to reference Markov model concepts that have been use in industrialized nation HAART cost-efficacy analyses such as the development of subway nations in capturing the quickly reducing use and mortality within the initial months of HAART treatment, and the availability of a Markov modeling state that reflects the stringent concept of HAART provision in South African general HAART policies.
The Markov model has in addition initiated the idea to capture the main cost-determiner of healthcare via transition cost, which is gained as a patient transits to death from Markov states. Probabilistic sensitivity analyses have acquired statistics indecision with 95/100 confidence interval regarding lifetime cost, results and ICER.
To conclude, the outcomes generated in this study indicate that HAART is a fairly cost-efficient technique to treat people suffering from HIV in relation to No-HAART. However, decreasing the costs associated with HAART would considerably enhance cost efficacy and permanent information regarding the comparable efficacy of HAART is needed so as to completely validate the outcomes of this study.
References
- Drummond M, Sculpher J, Stoddart G. Methods for Economic Evaluation of Health Care Programs. 3rd ed. Oxford: Oxford University Press; 2005.
- Drummond M, McGuire A. Economic Evaluation in Health Care – Merging theory with practice. Oxford: Oxford University Press; 2001.
- Miners A, Sabin C, Trueman P, et al. Assessing the cost-effectiveness of HAART for adults with HIV in England. HIV Medicine. 2001; 40(2): 52-58.
- Briggs A, Sculpher M. An Introduction to Markov Modelling for Economic Evaluation. Pharmacoeconomics. 1998; 13(4):397-409.
- Bozzette S, Joyce G, McCaffrey F, Leibowitz A, Morton S, et al. Expenditures for the care of HIV-infected patients in the era of highly active antiretroviral therapy. N Engl J Med. 2001; 344: 817–823.
- Gold R, Siegel E, Russell L, Weinstein C. Cost-Effectiveness in Health and Medicine. New York: Oxford University Press; 1996.
- Fox-Rushby J, Cairns J. Economic Evaluation. Oxford: Oxford University Press; 2005.
- Stoll M, Class C, Scuttle E, Graf M, Schmidt R. Direct costs for the treatment of HIV-infection in a German cohort after introduction of HAART. European J Med Res. 2002; 7: 463–471.
- Torres R, Barr M. Impact of combination therapy for HIV infection on inpatient census. N Engl J Med. 1997; 336: 1531–1532.
- Donaldson C, Mugford M, Vale L. Evidence-based Health Economics: From effectiveness to efficiency in systematic review BMJ Books, London; 2002.
- Freedberg K, Losina E, Weinstein M, Paltiel A, Cohen J, et al. The cost-effectiveness of combination antiretroviral therapy for HIV disease. N Engl J Med. 2001; 344: 824–831.
- Sendi P, Bucher C, Harr T, Craig B, Schwietert M, et al. Cost-effectiveness of highly antiretroviral therapy in HIV-infected patients: SWISS HIV Cohort Study. 2009; 13: 1115–1122.
- Badri M, Maartens G, Mandalia S, Bekker L, Penrod J, et al. Cost-Effectiveness of Highly Active Antiretroviral Therapy in South Africa. PLoS Med. 2006; 3(1).
- Beck E, Kennelly J, McKevitt C, Whitaker L, Wadsworth J, et al. Changing use of hospital services and costs at a London AIDS referral centre. AIDS. 1994; 8: 367–377.
- Beck E, Mandalia S, Williams I, Power A, Newson R, et al. For the NPMS Steering Group Decreased morbidity and use of hospital services in English HIV-infected individuals with increased uptake of anti-retroviral therapy 1996–1997. AIDS. 1999; 13: 2157–2164.