Effect of Vegetable Glycerin Concentration on Boiling Point
When the quantity of vegetable glycerin (VG) is increased in the e-liquid mixture, its boiling point increases. The thermal efficiency and the vaporization rate of VG /PG (vegetable glycerin /propylene glycol), change with their ratios. If the VG ratio in the binary e-liquid is high (at an ambient pressure and VG boiling point of 292°C and PG boiling point of 188.2°C), the mixture’s boiling point will be high (Duell et al., 2018).
The increased boiling temperature was observed because the liquid was allowed to remain in the liquid phase. An increase in the mole fraction of VG leads to a corresponding increase in boiling point; an increase in VG quantity leads to an increase in the mole fraction. In addition, the existing inter-molecular bonds, H-H bonds, in VG will need to be broken; therefore, the energy needed to break these bonds increases with temperature. An increase in VG liquid means an increase in osmotic pressure in the liquid, hence the continuous movement of molecules.
Volatility and Phase Behavior of VG/PG E-Liquid Mixtures
When a VG and PG mixture is boiled, the more volatile liquid is expected to evaporate while the less volatile liquid is expected to form the liquid phase. Propylene glycol is more volatile than Vegetable glycerin VG (Li, 2020). Above the boiling point line, a mixture of e-liquid and vapor and a pure phase are observed. An increase in temperature at constant pressure increases the formation of vapor. The composition of the substances in the two-phase region is at equilibrium.
From Figure 1, as the temperature increases, at a specific concentration of the mixtures, the liquid changes its phase from liquid to gas via an equilibrium phase of the liquid-gas phase. The PG/VG with a 30/70 ratio will have a lower boiling point, and the vaporization temperature is lower when compared with the 70/30 ratio. The e-liquid-vapor phase will form at a specific temperature and lies between the vapor and e-liquid phases.
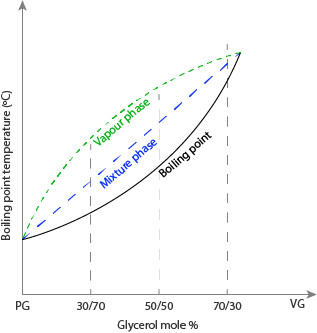
Liquid–Vapor Equilibrium in Binary E-Liquids
At the e-liquid and vapor phases, the compositions are at equilibrium. At equilibrium, the amount of e-liquid evaporating equals the number of molecules condensing into a liquid. That is, the rate of vapor formation equals the rate of condensation. Hence, the e-liquid and vapor phases are in equilibrium with each other.
Impact of Equilibrium Stress on Phase Shifts
When the equilibrium is altered, that is, the addition of liquid, it is said to be stressed. When the equilibrium is stressed, it shifts to the left or right. A system at equilibrium will shift when stressed, trying to undo the difference to offset the stress. An increase or decrease in substance in the equilibrium leads to a stressed system, leading to a phase transition (Cai, 2021). In Figure 2, the e-liquid and vapor mixture phase is at equilibrium before adding. The introduction of VG increases the boiling point; it is expected that the mixture phase will change to the vapor phase, but instead, it will shift to the liquid phase, shifting to the right.
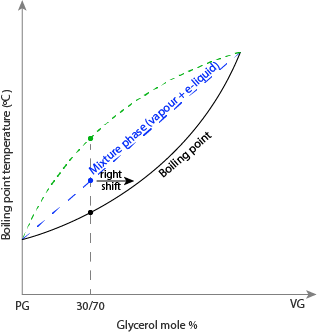
Thermodynamic Explanation of Boiling Point Elevation in VG/PG Systems
To conclude, the boiling point of the VG/PG mixture increases with an increase in the VG quantity because of an increase in osmotic pressure, an increase in concentration, and also the rise in the energy needed to break the H-H bond in the VG liquid. We have two phases during boiling: the e-liquid phase, the mixture phase (e-liquid and vapor), and the vapor phase. The vapor phase is at equilibrium, and when stressed, it shifts to offset the stress. Adding VG to the mixture causes stress to the equilibrium, shifting towards the liquid phase and to the right.
References
Cai, S., Wu, C., Li, X., & Li, Q. (2021). Effects of lubricant on evaporation and boiling processes of R1234ze (E): A molecular dynamics study. Applied ThermalEngineering, 193, 117009.
Duell, A. K., Pankow, J. F., Gillette, S. M., & Peyton, D. H. (2018). Boiling points of the Propylene glycol+ glycerol system at 1 atmosphere pressure: 188.6–292 C without andwith added water or nicotine. Chemical Engineering Communications, 205(12), 1691-1700.
Li, L., Lee, E. S., Nguyen, C., & Zhu, Y. (2020). Effects of propylene glycol, vegetable glycerin, and nicotine on emissions and dynamics of electronic cigarette aerosols. Aerosol Science and Technology, 54(11), 1270-1281.