Introduction
Milk is a nutritious, most-consumed animal product that contains proteins, minerals, vitamins, and carbohydrates. Milk sugar (lactose) is the primary source of disaccharide sugars, which have health benefits for human beings and animals (Marangoni et al., 2019). The lactase enzyme in the small intestine plays a crucial role in digesting lactose into its constituent monosaccharides, galactose and glucose, as illustrated in the figure below.
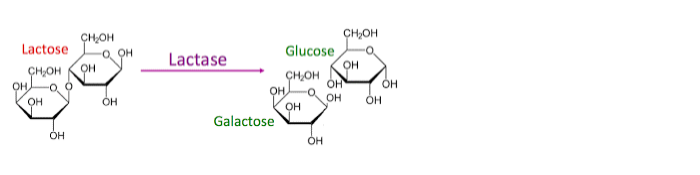
The breaking down of lactose and galactose is catalyzed by the lactase enzyme. According to Walker and Thomas (2019), after digestion, lactose is absorbed and used in the body. The inability to digest lactose in the villi leads to fermentation in the colon, producing fatty acids (lactate), hydrogen, methane, and CO2. Lactose intolerance is the inability to digest the lactose sugar in milk, and the resultant disease is called lactose malabsorption (Facioni et al., 2020). Individuals suffering from lactose malabsorption disease experience bloating, gas, and diarrhea after the consumption of dairy products.
Enzyme lactase (β-galactosidase) is, thus, industrially used to manufacture lactose-free dairy products, especially for people who exhibit lactose intolerance as a countermeasure to this condition. Lactase is mainly obtained from the fungus Aspergillus sp. and yeast Kluyveromyces sp. The Protease enzyme renin (chymosin) is responsible for the coagulation of milk proteins in the stomach through the hydrolysis process, as shown in Figure 2.

Coagulation of milk is vital to young animals, as it allows them to remain in the stomach longer for better digestion. Chymosin converts milk into a semi-solid curd and a liquid whey. This enzyme is usually the major protein component of milk casein (Mbye et al., 2021). This process is used in cheese making, where calcium ions are added to milk as calcium chloride (CaCl2).
Through this lab experiment, the process of creating lactose-free milk and the coagulation of milk is determined. The independent variables in this experiment are the type of milk (whole or lactose-free) and the CaCl2 concentration, while the dependent variable is the time to thickening and coagulation. The constant control variables included a temperature of 30°C, the time interval for observing milk thickening, the volume of milk, and the tube sizes.
Safety Measures
Students experimenting should wear personal protective equipment, such as a dust coat, because some reagents, such as calcium chloride, can cause desiccation and irritation. All reagents should follow spillage safety datasheets, where toweling and water should be used to wipe milk spills.
Apparatus
- 10 cm3 syringe.
- 50 cm3 of whole milk.
- 10 cm3 of 3% sodium alginate solution.
- 2x 100 cm3 beakers with 50 cm3 of 3% CaCl2 solution.
- 5 cm³ Lactozym (lactase enzyme).
- Tea strainer.
- Forceps.
- Distilled water in a wash bottle.
- Perfusion Column (Syringe barrel with a clamp at base).
- Large beaker.
- 4 glucose Diastix with access to scale.
- A 100 cm³ beaker on the bench.
- Paper towels.
- 12 sample tubes.
- Marker pen.
- 1 × 2 cm3 pipette.
- 1 × 0.1 cm3 pipette.
- Dropping pipette.
- 2 timers.
- 2 cm³ enzyme Chymosin.
- CaCl2 solutions of 0.05 M and 0.10 M.
Experiment
Method 1: Testing for Glucose Content
4 cm³ of Lactozym was drawn using the 10cm3 syringe, followed by the addition of 10 cm³ of 3% sodium alginate solution to the same syringe, and the syringe was shaken vigorously until the liquids formed a homogeneous mixture. 50 cm³ of a 3% CaCl₂ solution was added to an empty beaker, followed by the dropwise addition of an alginate enzyme mixture. The beads were left in the solution for three hours to settle and were poured out using a tea strainer, where they were rinsed thoroughly with distilled water.
The perfusion column was prepared using a Hoffman clamp, ensuring it was closed above a clean beaker. This step was followed by the addition of the beads to the perfusion column using forceps. Milk was slowly added, and the Hoffman clamp was adjusted to maintain a slow and steady milk flow rate. The treated milk was collected in a beaker, and its glucose content was tested. Glucose testing was performed on the treated milk twice, and the results were recorded.
Method 2: Measuring the coagulation and thickening time for milk with lactose and milk without lactose at different CaCl2 concentrations
Three tubes were labeled 0, 5, and 10, and 1.9 cm³ of whole milk was placed in these tubes using a pipette, followed by the addition of 0.1 cm³ of distilled water. The temperature was raised to 30°C, followed by the addition of a drop of the enzyme to the tube using a pipette, and the time was immediately started. The tube was rocked by turning it upside down while it was being held to maintain warmth. A sign of coagulation was checked every five seconds while the tube was held gently until thickening became visible. Time was recorded, and the procedure was repeated with CaCl2 and for lactose-free milk instead of whole milk.
Results
Discussion
The first section of this experiment involved treating milk and checking for glucose content. This step was achieved by adding enzyme/alginate in the presence of a CaCl2 solution. Alginate thickens milk by increasing its viscosity through the absorption of water molecules.
The addition of CaCl2 solution was used to test milk with calcium ions (Qin et al., 2020). Calcium ions raise the acidity in milk and strengthen protein molecules, causing them to form a thicker consistency. Diastix was used to measure the glucose level in the treated milk; thus, a high concentration of glucose indicated that the lactase enzyme had hydrolyzed lactose. The treated milk had a low glucose level (0.25%) compared to the untreated milk (1.86%).
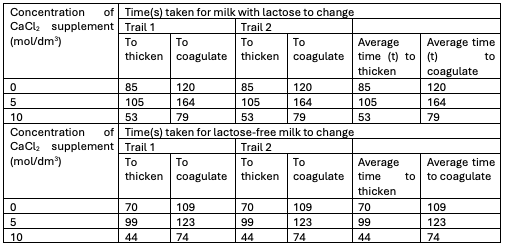
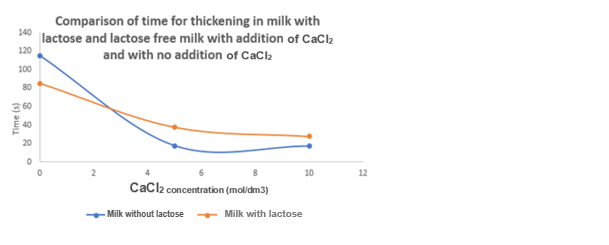
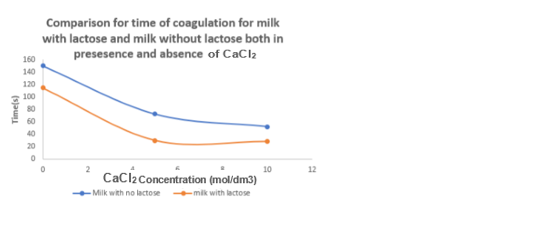
Part B of the experiment involved determining the thickening and coagulation times at different concentration levels of CaCl2. The results obtained from this experiment were graphed to facilitate a more comprehensive analysis. According to the findings, the addition of increasing amounts of CaCl2 resulted in a reduction of the thickening time for both lactose-containing and lactose-free milk (Olivares et al., 2022). The coagulation time of both types of milk exhibited this identical inverse trend.
CaCl2 is responsible for improving the recent coagulation process, which results in a shorter coagulation time being affected by variables such as chemical composition, somatic cell count, and chemical composition (Tarapata et al., 2020). From the result, it was observed that milk with lactose has a shorter time to thicken and coagulate than milk without lactose, as in Figures 3 and 4 above.
Evaluation
Sources of error in this experiment may have originated from contamination of the enzyme, systematic error, the use of larger beads, and the confusion between mL and cm³. The method of using a syringe and clamp was an inefficient way to obtain lactose-free milk, as it took a considerable amount of time to collect the treated milk. Students are required to wear gloves to prevent contamination of the enzymes. Moreover, if values are compared against the standards and using controls during the experiment, this can help avoid a systematic error.
Conclusion
The experiment was performed successfully despite the possibility of some sources of error. It was observed that the process of breaking down lactose during milk treatment for individuals with lactose intolerance is enhanced by the enzyme lactase. The experiment’s outcome was that a higher CaCl2 concentration shortened the thickening time for both milk types (lactose and lactose-free), and the coagulation time followed a similar diminishing trend.
It was also noted that the rennet coagulation process is improved by CaCl2, which shortens the coagulation time. Milk coagulation is affected by its acidity, chemical composition, and somatic cell count. In cheese making, CaCl2 is added because it promotes faster thickening and coagulation of the milk. The process of thickening and coagulation in milk is hindered by lactose. Safety measures include wearing gloves to avoid contamination, and systemic errors should be avoided by calibrating equipment and control experiments.
Reference List
Facioni, M.S., Raspini, B., Pivari, F., Dogliotti, E. and Cena, H. (2020). ‘Nutritional management of lactose intolerance: the importance of diet and food labelling.’ Journal of Translational Medicine, 18, pp.1-9.
Marangoni, F., Pellegrino, L., Verduci, E., Ghiselli, A., Bernabei, R., Calvani, R., Cetin, I., Giampietro, M., Perticone, F., Piretta, L. and Giacco, R. (2019). ‘Cow’s milk consumption and health: a health professional’s guide.’ Journal of the American College of Nutrition, 38(3), pp.197-208.
Mbye, M., Mohamed, H., Raziq, A. and Kamal-Eldin, A. (2021). ‘The effects of camel chymosin and Withania coagulants extract on camel and bovine milk cheeses.’ Scientific Reports, 11(1), pp.1-14.
Olivares, M.L., Costabel, L.M., Zorrilla, S.E. and de Vicente, J. (2022). ‘Calcium-induced skim milk gels: Effect of milk powder concentration and pH on tribo-rheological characteristics and gel physico-chemical properties.’ Food Hydrocolloids, 124, p.107335.
Qin, Y., Zhang, G. and Chen, H. (2020). ‘The applications of alginate in functional food products.’ J. Nutr. Food Sci, 3, p.100013.
Walker, C. and Thomas, M.G. (2019). ‘The evolution of lactose digestion.’ In lactose (pp. 1-48). Academic Press.