Abstract
Respiration is defined as the process of producing energy, typically through the absorption of oxygen and the emission of carbon dioxide, generated during the oxidation of complex organic compounds. Aerobic respiration is the breakdown of glucose in the presence of oxygen. At the same time, anaerobic respiration is the production of energy without the presence of oxygen.
Although both processes generate energy from glucose, anaerobic respiration typically yields carbon dioxide, ethanol, and energy, or energy and lactic acid, while aerobic respiration produces carbon dioxide, water, and energy. Thus, anaerobic respiration is crucial for humans, as it is used to produce alcoholic beverages. This process has been used in the production of alcohol since ancient Egypt.
This report describes an experiment conducted to compare the effectiveness of yeast fermentation among various sugars. The study measured the volume of carbon dioxide produced by different sugars, including glucose, fructose, sucrose, and starch. The analysis demonstrated that glucose produced the most significant amount of CO2, as it has the simplest molecular structure. Fructose produces less CO2, being an isomer of glucose with a slightly more complicated structure. Sucrose produced the third largest amount of CO2, while starch produced the least amount of CO2, as it is a polysaccharide.
Introduction
In general, respiration is understood as a process of energy production that typically involves the intake of oxygen and the release of carbon dioxide, which is produced through the oxidation of complex organic substances (Campbell & Paradise, 2016). Cellular respiration is a process that produces energy through the breakdown of glucose, typically via oxidation (Campbell & Paradise, 2016). There are two types of respiration, including aerobic and anaerobic. Aerobic respiration is a process during which glucose is transformed into energy, water, and carbon dioxide in the presence of oxygen.
There are four steps in the process, including glycolysis, the formation of Acetyl-CoA, the citric acid cycle, and the electron transport chain (Campbell & Paradise, 2016). Anaerobic respiration, or fermentation, is the process of breaking down glucose to generate energy in the absence of oxygen. While both processes produce energy from glucose, the end products of anaerobic respiration are typically carbon dioxide, energy, and either ethanol or lactic acid. In contrast, the end products of aerobic respiration are water, carbon dioxide, and energy (Campbell & Paradise, 2016).
Alcoholic fermentation is a biotechnological process used to transform sugars into ethanol and carbon dioxide. The process is usually accomplished with yeast or other kinds of bacteria. This process is used in the production of wine and beer (D’Amore et al., 1989; Wang et al., 2004). The history of alcoholic fermentation is deeply rooted in the history of our civilization, as evidenced by ancient Chinese and Egyptian records that document the use of alcoholic fermentation to produce alcoholic beverages (Banga, 2017). Two types of enzymes are responsible for alcohol fermentation, including invertase and zymase (Lodolo et al., 2008).
Several factors influence the activity of these enzymes, including temperature, pH, enzyme concentration, substrate concentration, and the presence of inhibitors or activators (Wu et al., 2018). Different sources of substrates, such as Starch, Sugar/Sucrose, Glucose, and Fructose, have varying effects on the rate of enzyme activity and fermentation. According to Quirao (2015), glucose, sucrose, and glucose help accumulate carbon dioxide, while starch, lactose, and distilled water do not accumulate CO2. According to Wang et al. (2004), glucose has the highest effect on the intensity of fermentation, with sucrose coming next, while fructose appears to be the least effective in making alcohol from yeast on a large scale. The products of alcoholic fermentation are ethanol, carbon dioxide, and energy, which are demonstrated in the chemical equation below (Campbell & Paradise, 2016).
C6H12O6 → 2C2H5OH + 2CO
Yeast is a unicellular fungus that is capable of producing alcohol. Saccharomyces cerevisiae is the most commonly used yeast in breweries and bakeries, as it is a top-fermenting yeast (Wang et al., 2004). Pyruvate Decarboxylase and Alcohol Dehydrogenase are commonly used enzymes in fermentation (Lodolo et al., 2008).
The purpose of this experiment was to determine how effectively yeast ferments in different sugars. Previous research has demonstrated that glucose is the most effective sugar in yeast fermentation, followed by sucrose, then fructose, and finally starch among the sugars. No fermentation is expected to take place in distilled water (Quirao, 2015; Wang et al., 2004). This experiment will utilize the net height change in carbon dioxide bubbles to test the hypothesis.
Methods and Materials
The study tested the effect of different sugars on the rate of carbon dioxide evolution. Five fermentation tubes were prepared that were later filled with solutions of 2% fructose, 2% glucose, 2% sucrose, and 2% starch. A container with distilled water and a container with yeast suspension were also prepared. Additionally, five smaller tubes were prepared, each containing a yeast suspension. The lab equipment included pipettes, glass rods, larger tubes, and a ruler. The setup of the experiment is provided in Figure 1 below.

Pipettes were used to add these substances to the smaller tubes for further experiments. A glass rod was used to stir the yeast suspension. After the larger tubes were placed on the smaller tubes and turned upside down, they were inserted into a large tube rack. A ruler was used to measure the initial height of gas bubbles. Respirometers were then put in warm water. After 20 minutes, the height of bubbles was measured and recorded, and the net change in the height was calculated.
Results
Five substances were tested, including fructose, glucose, starch, and distilled water. The height of bubbles was measured before and after the experiment. The experiment was designed to measure the increase in carbon dioxide bubble height after the bubbles were submerged in warm water at 32 °C for 20 minutes. The larger the change, the quicker the fermentation process was expected to go. The results are presented in Table 1 below.
Table 1: Collected lab data
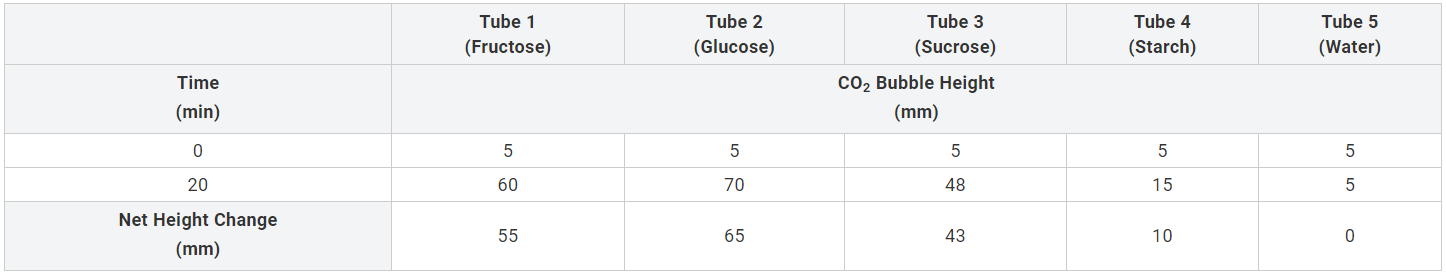
The results demonstrate that the height of CO2 bubbles was the same before the experiment. After the experiment, the heights of the CO2 bubbles were 60 millimeters for fructose, 70 millimeters for glucose, 43 millimeters for sucrose, 15 millimeters for starch, and 5 millimeters for distilled water. The data is visualized in Figure 2 below.
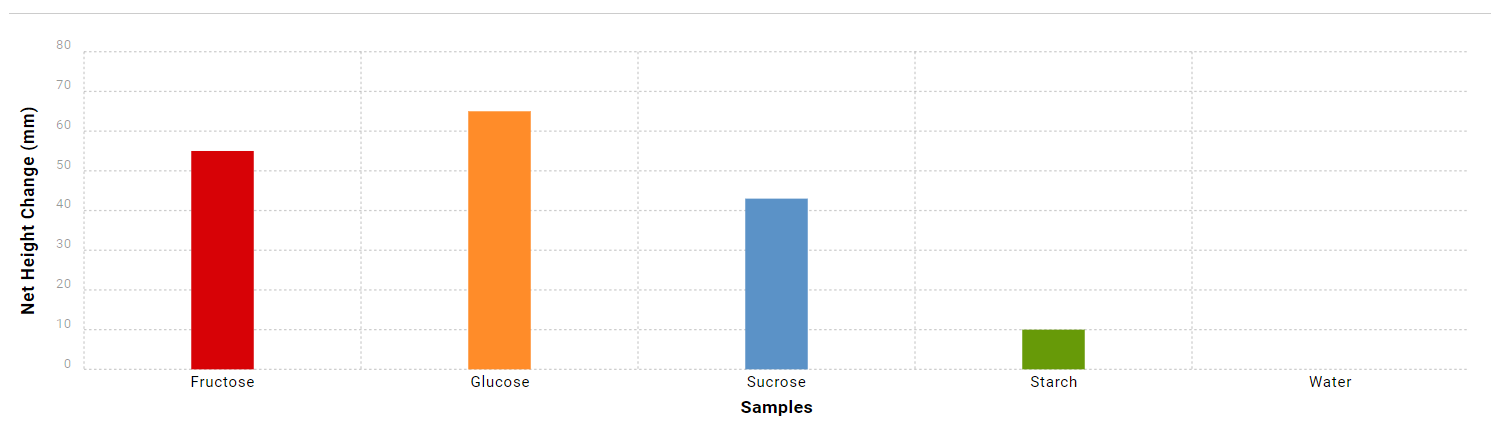
The net height change of CO2 bubbles was the highest for glucose, at 65 millimeters. Fructose was associated with a 55-millimeter increase in the height of carbon dioxide bubbles. Sucrose was associated with a 43-millimeter change in CO2 bubble height. Starch had the lowest effect on yeast fermentation, as the net change in the height of carbon dioxide bubbles was only 10 millimeters. Finally, yeast did not appear to ferment in the distilled water.
Discussion
The tube with glucose produced 65 milliliters of CO2 bubbles, as consumption of glucose was the fastest due to yeast being glucophilic (Wang et al., 2004). Fructose produced slightly less CO2 (55 mm), which was explained by a decreased rate of fructose consumption (Wang et al., 2004). Both glucose and fructose are monosaccharides, which makes their consumption of glucose/fructose almost the same (Wang et al., 2004).
However, since fructose is an isomer of glucose, its consumption rate was slightly different (Wang et al., 2004). Sucrose is composed of monosaccharide units, specifically α-D-glucose and β-D-fructose, which makes its consumption even slower (Quirao, 2015). As a result, it had a lower production of CO2 bubbles (43 mm). Starch is a polysaccharide comprising glucose monomers joined in α-1,4 linkages, which makes the fermentation process very slow (Banga, 2017). The analysis demonstrates that the larger and more complicated the molecules, the less CO2 was produced, which may imply that the speed of the reaction depends on the complexity of the substrates.
The results of this study confirmed the hypothesis that glucose would have the highest fermentation speed, followed by fructose, which would have the second fastest fermentation speed (Wang et al., 2004), sucrose would have the third fastest fermentation speed, and starch would have the slowest fermentation speed. The results of the experiment described in this report were in accord with previous research.
Conclusions
Glucose was found to be the most suitable substrate for yeast cells to ferment into alcohol. It can be used by combining the yeast with a 2% glucose solution and placing it in warm water. Glucose is the best for fermentation, as it has the simplest structure and can be easily decomposed. Future research can focus on testing the fermentation speed at different temperatures or using different sugars. Moreover, future research can use more concentrated solutions of sugars for analysis, which may help to refine the findings of this experiment.
References
Banga, J. (2017). Alcoholic Fermentation. Arcler Education Incorporated.
Campbell, A. M., & Paradise, C. J. (2016). Cellular Respiration. Momentum Press.
D’Amore, T., Russell, I., & Stewart, G. G. (1989). Sugar utilization by yeast during fermentation. Journal of Industrial Microbiology and Biotechnology, 4(4), 315-323.
Lodolo, E. J., Kock, J. L., Axcell, B. C., & Brooks, M. (2008). The yeast Saccharomyces cerevisiae–the main character in beer brewing. FEMS Yeast Research, 8(7), 1018-1036.
Quirao, H. A. (2015). The Effect of Different Sugars (starch, lactose, sucrose, glucose and fructose) To the Rate of Carbon Dioxide Evolution of Yeast in Fermentation. Web.
Wang, D., Xu, Y., Hu, J., & Zhao, G. (2004). Fermentation kinetics of different sugars by apple wine yeast Saccharomyces cerevisiae. Journal of the Institute of Brewing, 110(4), 340-346.
Wu, J., Elliston, A., Le Gall, G., Colquhoun, I. J., Collins, S. R., Wood, I. P.,… & Waldron, K. W. (2018). Optimising conditions for bioethanol production from rice husk and rice straw: effects of pre-treatment on liquor composition and fermentation inhibitors. Biotechnology for biofuels, 11(1), 1-13.