Background
Ascorbic acid, also referred to as vitamin C, is one of the essential nutrients in the human body. Vitamin C plays several roles in the human body, including collagen production, immune system function, and wound healing. Additionally, vitamins are crucial antioxidants that can protect cells from the effects of free radicals following food breakdown, as well as exposure to tobacco smoke and X-rays or other sources. Vitamin C is a crucial component because it has several health benefits related to human health, and its deficiency can lead to scurvy. The nutrient is found in several fruits and vegetables, including the juices of citrus fruits, tomatoes, and potatoes.
Aim
The purpose of this experiment is to determine the concentration of ascorbic acid in various fruit juices through titration with a similar titrant, 2,6-dichlorophenolindophenol (DCPIP). The endpoint is considered to have been reached when the juice reduces the DCPIP to a colorless or faint pink solution. The concentration of ascorbic acid varies among different fruits and may be influenced by factors such as storage conditions, measurement procedures, and juice processing procedures. The purpose of this experiment is to measure the ascorbic acid content in various fruit juices using titration.
Materials & Methods
Materials include measuring cylinders, volumetric flasks, burettes, pipettes, conical flasks, test tubes, and reagents, including DCPIP solution, standard ascorbic acid solution, and a metaphosphoric-acetic acid mixture (MPA). Standardization of the dye can be achieved by titrating a specific concentration of ascorbic acid with DCPIP to determine the equivalent quantity of ascorbic acid per 1 cm³ of dye.
Results
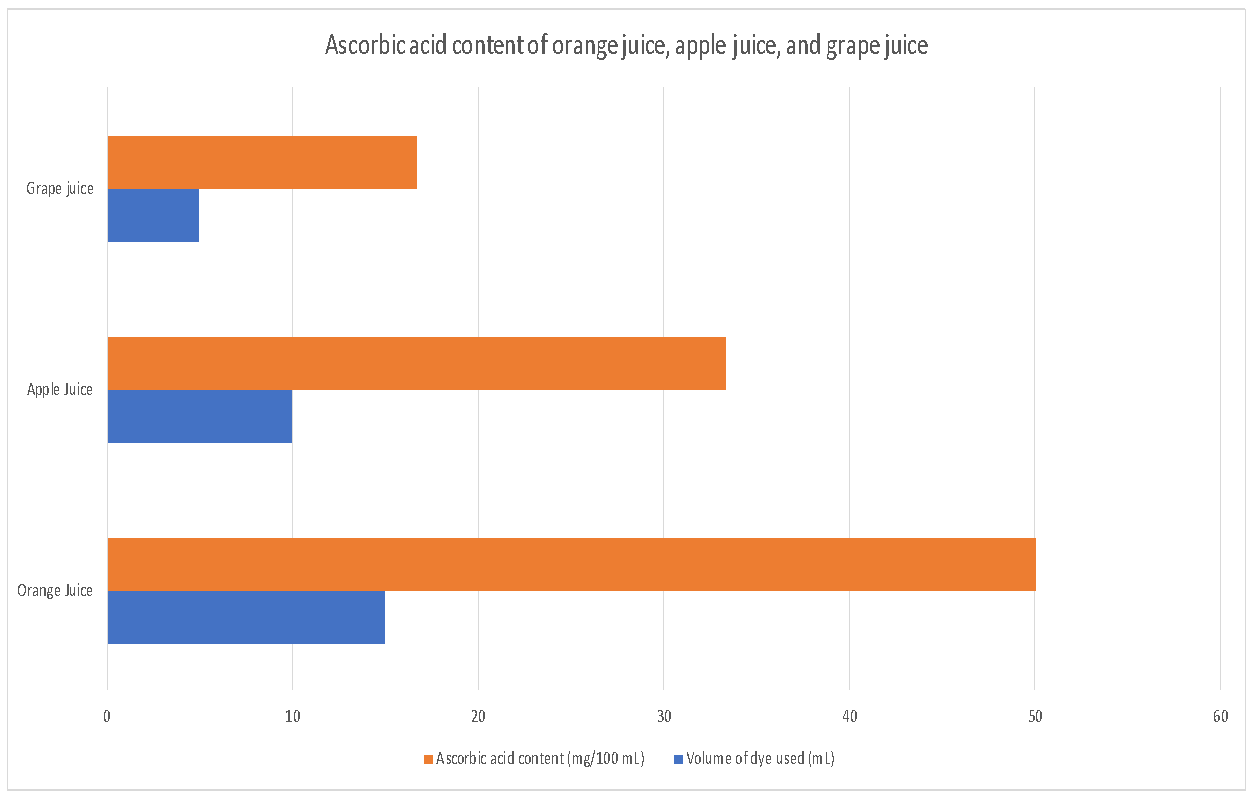
The juices tested were apple, orange, and grape, and the protocol procedure was followed. The orange juice had 50 mg/100mL, while the apple juice had an acid content of 33.33 mg/100mL. Grape juice had an acid content of 16.7 mg/100mL. It is observed that orange juice has the highest ascorbic acid concentration, followed by apple and grape juice. In the graph below, the volume of dye is represented in comparison with the concentration of ascorbic acid in each fruit. The trend increases from grapefruit to apple juice, and then to orange juice.
Discussion
The calculation of the ascorbic acid was performed by dividing the volume of the dye by the volume of juice used. For instance, the ascorbic acid content in orange juice is calculated by dividing 15 mL by 5 mL, resulting in 50 mg/100mL. The results indicate that orange juice has a higher ascorbic acid content compared to other fruit juices tested. These results are consistent with previous studies, which have tested the content of ascorbic acid in fruit juices (Soural, Šnurkovič, and Bieniasz, 2022, p. 13). The possible sources of error include inaccurate measurement of the fruit juice and dye solution volumes, incorrect identification of the endpoint, and the presence of reducing agents within the juices other than ascorbic acid.
A potential limitation of this experiment is that the method is not ideal for solid samples. The procedure only measures total ascorbic acid, as opposed to specific forms of vitamin C. Biosensors and high-performance liquid chromatography (HPLC) can be used to enhance the analysis of ascorbic acid by increasing efficiency and accuracy (Sani et al., 2023). HPLC is more accurate and sensitive than DCPIP, while biosensors can detect and measure individual substances, including different forms of vitamin C.
Reference List
Sani M. A. et al. (2023) ‘Metal–organic framework fluorescence sensors for rapid and accurate detection of melamine in milk powder‘, Biosensors, 13(1), pp. 1-16.
Soural, I., Šnurkovič, P. and Bieniasz, M. (2022) ‘Chemical comparison of 100% apple, orange and grapefruit juices directly pressed and made from concentrate‘, Czech Journal of Food Sciences, 40(1), pp. 69-75.