Background
Marfan syndrome (MFS) is a rare kind of inherited disease prevailing in 1/5,000 individuals. The diagnosis of MFS is sometimes difficult due to several signs and symptoms on diverse organ systems that require consultancy of different specialists. The clinical criteria based on prevalence mainly lie in skeletal-ocular-cardiac systems of the patients. Among many skeletal abnormalities, Dolichostenomelia is characteristic of excessive growth of limbs and absence of parallel development of muscle and fat. The patients of all age groups in 90% cases display features of being excessively thin and tall with broad arm span (arm span: height ratio >1.05:1, while in normal healthy person it is close to 1:1). Over 60-80% patients display signs of eye lens and/or retinal disorders, mainly the dislocated lens. Consequently, about 40% patients suffer from shortsightedness (myopia). The other problems are associated with cardiac involvement and about 90% of MFS patients die of cardiovascular complications mainly the aortic rupture. The clinical diagnosis becomes obvious if the patient has a family history of the disease. MFS is recognized as autosome-linked inheritable disease due to a mutation in MFS1 gene localized in chromosome #15. This defect hampers Fibrillin-1 protein synthesis responsible for assembly and organization of fibrin microfibrils, a constituent of connective tissues. MFS also exhibits genetic diversity and a second gene, called MFS2 was localized in chromosome #3 coding for some unknown protein. Genetic aberration in this gene accounts of about 8-15% of MFS. As a genetic counselor, I am presented with a case of Anne (16 years) with a suspicion that she might have MFS. After initial diagnosis her physician advised her to consult a Genetics Counseling Clinic.
Problem Question
Is it likely that Anne has MFS? It is hypothesized that she has. This is because her clinical symptoms like very tall at her age, nearsightedness or myopia, and most importantly arms pan: height ratio of 1.08:1, which is significantly higher than an average individual. However, it is also likely that height and myopic conditions are coincidently present and have nothing to do with MFS. Having only one clinical attribute (higher arms pan: height ratio) may not be indicative of MFS.
Procedure
For a better insight into Anne’s conditions it was necessary to work on the family history of MFS and for this purpose her pedigree chart was brought and for each member of the family (siblings, parents, siblings of mother and father, maternal and paternal grandparents) information on the clinical symptoms of any past or present disease condition and cause of death were collected. In pedigree chart males were designated by square and females circle signs. The two generation hierarchies above and one generation hierarchy below the Anne’s generation was pooled and presented in the chart as below:
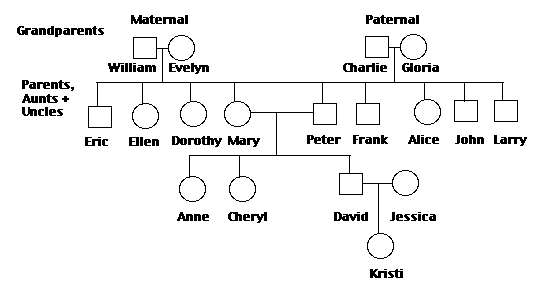
Results
Firstly, the typical clinical symptoms attributed to MFS were sorted from the description of living and deceased members of the family. A single symptom and symptoms that can be due to other causes were sorted as possible mild MFS. When two or more atypical symptoms and/or only one typical symptom were present, the cases were considered as being mild to severe and if more than one typical symptom were present they were recognized severely affected patients. There was no quantitative data available on the magnitude of problems and thus the data were generated from given qualitative information. The following table illustrates presence of MFS symptoms in generation hierarchies down the order:
There were other clinical symptoms but they were not associated with a possibility of MFS and were discarded. With respect to MFS these individuals were considered as being healthy. From the data it appears that most likely Anne’s paternal grandfather Charlie had this disease and her father Peter and Uncle John inherited it. Frank, Alice and Larry were apparently free of the disease. In her generation, David and she inherited the father’s disease allele and surprisingly even Cheryl displayed some mild symptoms. One bottleneck in analysis is that Anne’s parents and grandparents had miscarriages and some of the relatives died young and hence the symptoms of those individuals could not be recorded in the pedigree chart. The data also suggests that Anne did not inherit the disease from maternal (Mary’s) side and none of the blood relations of mother had any MFS symptom. A significant feature of the inheritance pattern is that as generations proceeded more family members acquired the disease. The disease acquisition was not restricted to any particular sex. Kristi, nice of Anne was too young to present any symptom of the disease (if any).
Analysis
From the initial clinical symptoms Anne certainly has MFS. In order to verify we take a help of genetic analysis and inheritance of dominant autosomal allele, MFS1. The chances of getting dominant allele in F1 is 50% (1/2), means if there are four children two are expected to get the allele and disease. If more than four then the fifth child may have 50% chance of getting the disease. The following Punnett’s squares show the possible gamete combination for dominant autosomal MFS1 allele down the generations assuming that dominant MFS allele is M and the recessive normal allele is m:
Paternal Grandparents
Parents
From these Punnett’s squares it is clear that Charlie was heterozygous for MFS allele and it was segregated in two children Peter and John whereas others were homozygous for the corresponding recessive normal allele and they were disease free. Likewise, in next generation Peter was heterozygous for MFS allele and it was segregated in Anne and David and they would show up the disease. Surprising homozygous for recessive allele, Cheryl also displayed some mild symptoms. The likely explanation for Cheryl is that recombination between MFS1 and normal alleles took place before segregation. Alternatively, the second gene MFS2 might have mutated and manifested as secondary trait of MFS. The pedigree chart would now indicate disease flow as follows:
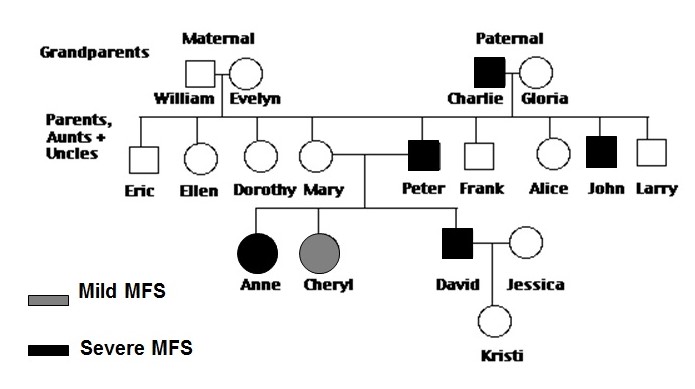
Conclusion
Both clinical and genetic data supports the hypothesis that Anne is suffering from MFS. She has inherited this trait from her father and him, in turn, from his father. The disease traits were not transmitted from maternal side. The only bit of uncertainty whether Anne inherited the disease or not comes from the fact that her two siblings died unborn. Had they been living and inflicted the disease her chances were almost nil to get the disease. This is because already David and the unborn children had the disease leaving no possibility for Anne to acquire the MFS1 allele, as it can not be more than ½ in F1. As such Cheryl’s case is also doubtful. As a genetic counselor and with all ethical and professional responsibilities I would advise Anne to get further check-up for MFS and make MFS1 RFLP fingerprint for verification. Perhaps she should also advise David (brother) to make sure that after one miscarriage Kristi has a strong chance to inherit fathers MSF1 allele and likely to show up the disease at later age. Cheryl’s problem is not grave and she can take medical help to rectify it. In long term Anne should be cautious about raising children because 50% of them might have a chance to acquire MFS. Overall, this kind of genetic counseling opens up new avenues to genetically diagnose the disease causing autosomal recessive and dominant alleles as well as sex chromosome linked inheritable alleles. Autosomal dominant MSF1 in particular has more prevalence in F1 compared to the other disease alleles and hence more rigorous pedigree analysis is desirous for Anne and many others who are suspicious of having clinical symptoms of this disease.
Discussion
To sum up, genetic counseling is a great option to decipher causes of disease symptoms and deaths in earlier generations somehow missed in clinical terms. More importantly it can give advice to prenatal individuals whether the unborn child may have disease gene or not, giving an option for parents to terminate pregnancy if desired. Some of the inherited diseases like muscular dystrophy are congenital and show up only at late age.