Executive summary
This paper is aimed at developing an action plan for such company as Eli Lilly. First, it will explain the nature of difficulties faced by this pharmaceutical corporation. Secondly, it will evaluate various marketing decisions which the company could take under those circumstances. It should be noted that this analysis relies primarily on the information presented in the case study by Elie Ofek and Ron Laufer (2008).
This case study focuses on the strategy or strategies that this organization had to choose in 2000. They were primarily related to the development of such a medicine as duloxetine or Cymbalta. The main recommendation that one can make is that this pharmaceutical corporation should attach greater importance to their brand image.
In particular, product development efforts and marketing campaign should first emphasize the efficiency of their drug as an antidepressant and tolerability. Its analgesic capacity should be a supplement but not the main attribute of the product. The rationale for these recommendations will be explained in the following sections of the paper.
Major issue
The problem faced by Eli Lilly had several dimensions. First, one should say that for a long time this pharmaceutical company was the major supplier of selective serotonin reuptake inhibitors (SSRI), especially fluoxetine, commonly known as Prozac. They were the only company that could sell the antidepressants based on this chemical molecule that they developed.
However, this situation could significantly change due to patent expiration. The thing is that manufacturers of generic drugs could bring very similar and less expensive products into the market, and it would have been difficult to compete with them only by means of price differentiation (Ofek & Laufer 2008, p. 1).
Hence, this organization had to introduce a new product that could successfully replace Prozac. At that point, the only antidepressant that could do that was Cymbalta or duloxetine (Ofek & Laufer 2008, p. 8). Overall, this drug had some significant strengths and limitations. It could treat various forms of depression and pain that sometimes accompanied this psychological disorder.
Nonetheless, it required a higher dosage, especially in comparison with Prozac or other antidepressants. At that point, they did not know whether QD (QD means once daily. This abbreviation is derived from Latin expression quaque die) dose of Cymbalta could effectively treat major depressive disorder (MDD). The management of Eli Lilly had to choose between several options.
On the one hand, they could invest into a clinical trial that could prove the efficiency of Cymbalta in treating MDD. On the other hand, they could carry out a clinical investigation which would demonstrate that this antidepressant could effectively mitigate the pain of the patients with mood disorders (Ofek & Laufer 2008, p. 8).
Therefore, this organization had to take a crucial marketing decision. They could emphasize the overall efficiency and safety of their product. Yet, they could also lay stress the idea that it could create extra value for patients. Another issue that one has to consider is time and cost constraints.
The thing is that the generic versions of Prozac could be introduced within the next two years or even less. In turn, a large scale medical trial could last for approximately 16 month and its average cost ranged from $25 to $50 million (Ofek & Laufer 2008, p 2). The decision that the management was to take had to account for these time and cost constraints. These are the main dimensions of the problem that Eli Lilly had to resolve.
Alternative solutions
This section of the paper will evaluate various options which this organization had at that time. First of all, they could emphasize on the efficiency and high tolerability of their product. Hence, they could first invest into a large scale clinical trial that can prove that 60 mg. of Cymbalta administered once a day could help the patients suffering from depression.
Moreover, this clinical trial had to demonstrate that this antidepressant had a very high tolerability rate. This aspect is probably even more important that overall efficiency of the drug. The thing is that many physicians attached and continue to attach greater importance to tolerability of medication (Ofek & Laufer 2008, p.12). Their preferences can be better illustrated by means of this diagram.
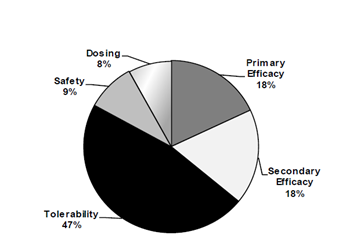
(Ofek & Laufer 2008, p.24)
Thus, by pursuing this strategy, Eli Lilly could win the trust of physicians. Nonetheless, this approach has some significant drawbacks. The problem is that Eli Lily’s competitors were also likely to place emphasis on such properties of their products as efficiency and high tolerability.
Moreover, their products could have been less expensive than Cymbalta. Therefore, there was great likelihood that Eli Lilly could lose competitive advantage to other pharmaceutical companies.
There was another alternative which was available to this organization at that moment. They could demonstrate that their drug could treat other comorbidities of the patients, especially pain. For instance, they could conduct a clinical trial showing that Cymbalta could be used as a reliever of diabetic peripheral neuropathic pain or headache (Ofek & Laufer 2008, p. 14).
Admittedly, such an approach could enable them to differentiate their product among other. In this way, they would be able to meet some extra needs of their patients. Nonetheless, this approach has several disadvantages. First, Cymbalta could be effectively marketed if they could be administered on a once-in-a-day basis.
In other words, if this drug had to be used only twice a day, many physicians could give preference to other antidepressants (Ofek & Laufer 2008, p. 1). Furthermore, this drug could not be marketed only as a pain reliever, because there were virtually thousands of other medicines which could be used to alleviate various forms of pain.
Thus, the new product of Eli Lilly could hardly compete with them. This is the main drawback of this strategy. It is quite probable that it could lead to disastrous outcomes, especially failure to meet the key needs of the patients.
The third method is to postpone the release of duloxetine and conduct every necessary clinical trial prior to submitting this antidepressant to Food and Drug Administration. In this way the company would have been able to introduce a dramatically new product to the market. The physicians as well as customers would have easily been able to differentiate it among others.
However, as it has been noted before a clinical trial can last for 16 months. Thus, the company will have to delay the release of Cymbalta for three or even four years. During that time, other manufacturers of anti-depressants could introduce their products. This competition could weaken the positions of Eli Lilly.
Overall, one can see that each of these strategies has its strengths and weakness. Yet, we can single out three distinct risks that they need to avoid, namely lack of differentiation, lack of focus, and significant delay in release. None of these risks could be neglected by the management. The following section will present some suggestions that can be followed by this company.
Recommendation
The recommendations proposed in this paper will be based on the premise that Eli Lilly is known primarily as “the Prozac Company” (Ofek & Laufer 2008, p.9). They are famous mostly for the treatment of depression, while painkillers or analgesics are not their specialty. It is quite likely that they were not able to compete with companies focusing on therapeutic area.
These organizations as well as their products already established a reputation among physicians and patients. Therefore, while developing and marketing Cymbalta they should first set stress its anti-depressive quality. In other words, this organization has to rely on its brand image which is one of their major assets, along with R&D.
This is why the company must conduct those medical trials which can prove is more efficient than Prozac, placebo, or any other medications which are often prescribed by psychiatrists. To some degree, this recommendation is supported by preliminary studies which showed that the patients who were given duloxetine, displayed fewer signs of depression (Ofek & Laufer 2008, p. 23).
Their advertisements must focus on the idea that this drug is a direct successor of Prozac and that it can be superior to its predecessor. If the managers of this company choose to act in this way, they will differentiate duloxetine among other antidepressants.
The second important recommendation refers to tolerability. The main task of Eli Lilly is to show that duloxetine not only treats various forms of depression more efficiently, but that its hypothetical complications are less frequent and dangerous.
Provided that the company manages to demonstrate this improvement by clinical trials, they will be able to retain their leading positions. Their success will be based on several pillars, namely, their brand name, efficiency of their product, and its safety.
When they receive the approval of Food and Drug Administration, they will be able to market Cymbalta as an antidepressant. When this product receives a certain share in the market, Eli Lilly can conduct a clinical trial demonstrating that this drug can be useful to the patients suffering from chronic pain. Thus, in this way the company will be able to increase the scope of the product and subsequently increase its market share.
Plan of action
On the basis of these recommendations it is possible to single out specific steps that Eli Lilly should take:
- Conduct a clinical trial proving that a once-in-day dose of duloxetine can be efficiently treat different forms of depression such as Major Depressive Disorder (MDD) or chronic depression;
- Submit the results to Food and Drug Administration;
- Launch an advertising campaign which will lay stress on the safety and efficiency of the drug. These qualities are more likely to appeal to the customers.
- Carry out the second trial that can demonstrate the efficiency of Cymbalta as analgesic.
- Start a new marketing campaign which would argue that this medicine can fulfill several functions such as mitigation of depressive symptoms and alleviation of pain. Hence, those people, who suffer from depressive disorders, will feel safe to use it as a painkiller.
At this moment, it is rather difficult to determine how much it will take the company to implement the plan. Given that a large clinical trial lasts for approximately 16 or 18 moths, one assume that the company will have to spend at least three years. Moreover, one should take into account that Cymbalta will not be able to win the trust of physicians and patients at once.
Even if the medicine proves to be as effective as it ought to be, it will get a considerable market share only in a year or even more. Hence, Eli Lilly will be able to implement this plan in no less than four years.
By adopting this plan, the management can ensure that the product addresses the needs of the patients and creates extra value for them. This is its major advantage of this approach. This solution appears to be the most optimal one given the competitive environment of Eli Lilly and that the products that it offers.
Expected outcomes
This plan can bring the following benefits to this pharmaceutical company:
- increased affiliation of their clients;
- a larger market share for the product;
- higher return on investment;
- increased profitability.
Apart from purely quantitative benefits, this strategy will let the company gain practical experience in therapeutic area. Moreover, this approach can minimize some of their risks such as failure to address the needs of the target audience or lack of differentiation. The main advantage of this strategy is that it utilizes the main strengths of Eli Lilly.
Reference List
Ofek, E. & Laufer, R. (2008). Eli Lilly: Developing Cymbalta. Boston: Harvard Business Publishing.