Introduction
Nowadays, there have been a lot of concerns with respect to the use of alternative sources of energy. Such concerns have resulted from the fact that most of the commonly used sources of energy have numerous consequences. For example, the burning of fossil fuels leads to the emission of greenhouse gases, which have enormous impacts on the environment. For this reason, there is a need for the adoption of sustainable sources of energy. Ethanol is considered a better alternative source of energy. Various locally available materials can be used to produce ethanol. Some of the commonly used materials include corn, bio-based feedstocks, sugar cane, petroleum, as well as algae. This paper provides a comparative analysis of the various materials used in the production of ethanol.
Background
Ethanol is a colorless liquid that is both flammable and volatile. It can be used as both an alcoholic beverage and as an alternative biofuel. It is easy to manufacture as well as process ethanol through a process similar to the one used in the production of alcohol. One advantage of the use of ethanol as a source of energy is that large amount of it can be produced over a short time. In addition, its production is not complex as it involves the use of crops such as sugarcane or corn. For this reason, this type of biofuel is renewable. In the United States of America, ethanol has been adopted as a suitable alternative source of energy. Such an approach was attributable to the fact that using ethanol fuel can help in the reduction of oil imports and the provision of the necessary support to the agricultural sector by encouraging the growth of corn and other crops that can be used in the production of ethanol.
In addition, the use of ethanol as fuel can be very instrumental in the reduction of the environmental impacts of using other types of fuels. For example, the United Kingdom has been working hard to reduce the consumption of fossil fuels based on the country’s strategy to adopt renewable energy sources for a chance to reduce the amount of harmful emissions to the environment. In spite of the significance of ethanol as fuel, its production is limited due to the lack of enough land to grow the crops used in its production.
Analysis
As pointed above, ethanol can be produced from corn, sugar cane, algae, petroleum (hydrolysis of ethylene), or even cellulose. The process of producing ethanol using each of the above materials is described below.
Ethanol production using corn
The United States of America is one of the major producers of corn in the world and hence, uses such opportunity to produce ethanol fuel. The initial process of making ethanol from corn starts with the selection of the right quality of corn for use in the fermentation process. The corn is milled to form grain-like powder through either dry milling or wet milling. The fine powder obtained is allowed to mix with enzymes and waster to enhance the formation of a slurry. The role of the enzymes at this stage is to help in the conversion of the available starch to dextrose, which is a simple sugar. The level of the PH is controlled through the addition of ammonia before the mash is heated and cooled to allow yeast to work on the sugars present to form ethanol. The diagram below provides a summary of the ethanol production process using corn.
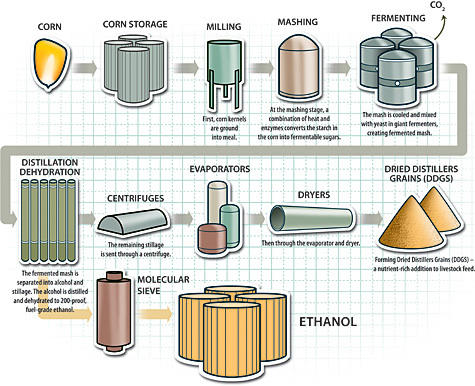
The use of corn in the production of ethanol has a number of drawbacks. For example, there are concerns over the energy return available when ethanol is produced from corn. The production of ethanol from corn is associated with negative impacts on the environment. This is attributed to the fact that the production of high amount of ethanol requires corn in large quantities. For this reason, such production of corn has a high probability of damaging the environment due to the approach used in growing this type of crop. The “fencepost to fencepost” way of planting subjects the soil to massive erosion. Additionally, the production of corn causes drop in the water level due to the fact that high quantity of water is used.
Secondly, the production of high quantities of ethanol from corn is subject to mandates as well as subsidies. The implication is that the price of corn ethanol can be quite high relative to the subsidies and labor required.
Thirdly, the use of corn in the production of ethanol fuel requires a lot of resources including some which could be useful in the production of food. Such a scenario triggers increases in the prices of food this is attributable to the fact that the world is facing problems of food shortages. As such, any available resources ought to be channeled towards the production of food rather than being spent producing ethanol that is not economical.
Ethanol production using sugar cane
The primary feedstock used in the production of ethanol is sugar cane molasses or the juice. These are by-products obtained from sugar milling factories and hence, make the production and use of ethanol sustainable. India relies heavily on sugar cane molasses for the production of ethanol. On the other hand, fresh sugar cane juice is commonly used in the production of most ethanol in Brazil.
The production of ethanol from sugar cane involves the extraction of sucrose from cane that has been cut and milled to produce juice. The clarification step involves hating of the juice to about 115°C, treating it with sulfuric acid and lime for the purpose of removing any unwanted inorganic materials. This step is followed by the fermentation of the juice and molasses to obtain about 10-20% sucrose. During this stage, the reaction is maintained at fermentation conditions by cooling the mixture before adding yeast.
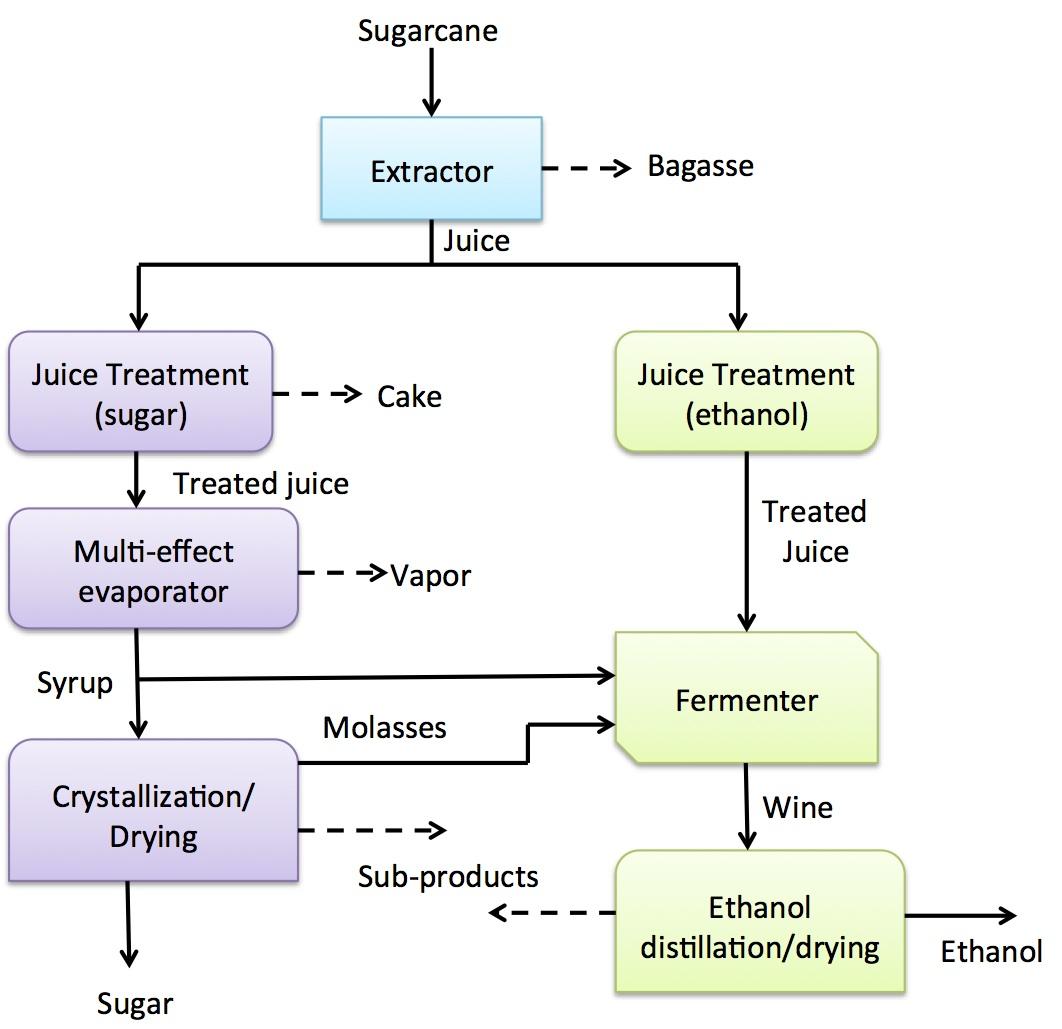
Advantages of using sugar cane to produce ethanol
The production of ethanol from sugar cane has both advantages and disadvantages. Some of the advantages are highlighted below. First, sugar provides a significant balance in terms of greenhouse gas emissions during the production and use of ethanol. Past studies have showed that the production of biofuel form sugar accounts for above 80% of greenhouse gas reductions when compared to the used of fossil fuel. When compared to ethanol from corn, sugarcane ethanol is 35% better in terms of avoided emissions.
Secondly, given the limit in terms of land to growth crops for the production of ethanol, a high yield of ethanol can be produced from sugarcane planted in a small piece of land. Sugarcane ethanol is considered highly productive when compared to other sources of ethanol such as corn. This is attributable to the superior sucrose yield from sugar juice of molasses. Additionally, the ethanol production from sugarcane contains the highest energy balance based on the amount of waste renewed in comparison to the energy inputs. For example, the energy balance of ethanol produced in Brazil from sugar is 9.0, while the ethanol produced form American maize has an energy balance of 2.0. Generally, sugarcane is considered to be an appropriate alternative for bio-fuel production with respect to environmental, energy, as well as economic impacts.
Disadvantages of using sugar cane to produce ethanol
The production of ethanol from sugarcane faces a number of challenges that affect its efficiency as an alternative source of biofuel. First, the suitability of land for use in planting sugarcane is limited. This is especially the case for industrialized countries like China, since it is impossible for them to expand the existing land to cater for the production of sugarcane. In addition, harvesting the planted sugar is also a challenge due to the fact that such process requires a lot of investment in terms of capital and machinery. This challenge is most common for countries whose terrain is hilly since such type of lands requires the implementation of mechanized harvesting. On the other hand, cost and benefit issues are likely to affect the expansion of land for the production of sugar. This is attributable to the fact that such expansion can be labor and capital-intensive.
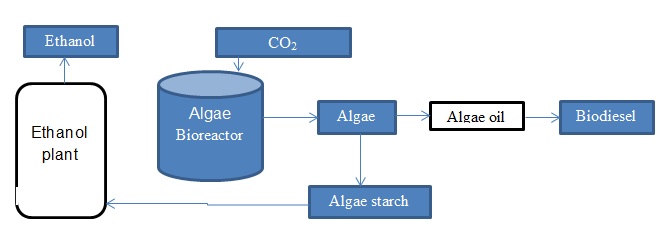
Production of ethanol from algae
Algae can be described a simple organisms that contain chlorophyll and have the ability of growing both heterotrophically and phototrophically. In the case of the phototrophic growth of algae, the carbon dioxide available in the atmosphere is converted to carbohydrates. There ae two classes of algae which include the macro-algae and microalgae. Algae can be used in the production of bioethanol and biodiesel. The micro-algae are a suitable feedstock in bioethanol production due to its capacity to produce lipids, as well as it has high photosynthetic efficiency.
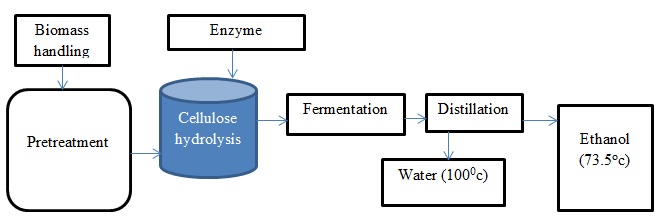
Ethanol production using cellulose
The production of ethanol through the cellulosic process comprises of the pretreatment stage, the hydrolysis of enzymes, fermentation, as well as distillation. The first stage is used to ensure that the cellulose is highly susceptible to breakage as a preparation for the hydrolysis of enzyme. It involves the removal of hemicellulose and lignin thereby exposing the inside part of the cell wall. The enzyme hydrolysis follows the pretreatment stage to enhance the conversion of polysaccharides to simple sugars that can be acted upon by yeast or bacteria. The third stage is the fermentation process where single sugars are converted into fuel ethanol. In this case, the major organisms used in the conversion of sugars to ethanol include bacteria and yeast.
Ethanol production using petroleum
Ethanol can also be produced from petroleum through the hydrolysis of ethylene. This is achieved through the reaction of ethylene with steam in an exothermic and reversible reaction.
Ethylene is passed through a reactor containing steam that converts about 5% of it every time the substance is passed via the reactor. This process is successful due to the fact that the reaction between ethylene and steam is reversible. The implication is that the hydrolysis of ethylene can lead to 95% conversion since ethanol is constantly removed from the equilibrium. The flow chart below provides a summary of the formation of ethanol from ethylene.
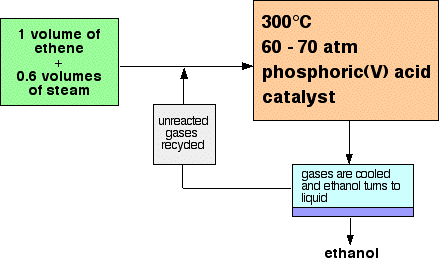
The gases from the reaction undergo a cooling process to condense ethanol and any excess steam. After condensation, fractional distillation is used to separate water from ethanol. The use of the fractional distillation method is appropriate at this step because water and ethanol have different boiling points making it suitable for the liquids to be separated based on the fact that ethanol will distil off first (at 73.5°C).
While the production from ethylene is simple and easy, it involves the use of high pressures which are quite costly due to the numerous requirements involved. On the other hand, such pressures cause the polymerization of ethane making it difficult to produce ethanol of the required quantity.
Synthesis of Ethanol (Reactions)
The production of ethanol fuel comprises of three process including fermentation, distillation, as well as dehydration. However, the primary process of producing ethanol can be described in three steps: photosynthesis, fermentation, and combustion as shown in the reactions below.
Photosynthesis
6CO2+6H2O+light=6C6H12O6+6O2
Fermentation
6C6H12O6 = 2C2H5OH+2CO2 + heat
Combustion
C2H5OH = 2CO2+3H2O + heat
The addition and balancing of the three equations gives ethanol. During the photosynthesis reaction, light is converted to heat. In this step, the production of ethanol involves the fermentation of sugars by microbes. The second stage involves the removal of water from the ethanol. However, it is important to limit the purity of ethanol at 95%duirng the distillation stage to avoid forming water-ethanol azeotrope. The dehydration step is used primarily to remove any water molecules left.
Separation of Ethanol
The separation of ethanol based on the principle of separating liquid-liquid mixtures. As such, for the separation to be achieved, the liquid compositions should be immiscible. In most liquid-liquid extractions, the miscibility of the solvent is a significant factor of consideration. In the case of ethanol, its separation follows the principle of distillation where the process is possible due to difference in the boiling point of water and ethanol.
E-factors of Ethanol Production
The use of corn in the production of ethanol fuel requires a lot of resources including some which could be useful in the production of food.
On the other hand, the suitability of land for use in planting sugarcane is limited. This is especially the case for industrialized countries making it environmentally-demanding to expand the existing land to cater for the production of sugarcane.
Even though, the production of ethanol from ethylene is simple and easy, it involves the use of high pressures which are quite costly due to the numerous requirements involved, as well as has a high percentage of waste materials released to the environment.
Conclusions
Ethanol has been considered to be an appropriate alternative source of renewable and sustainable energy. With the increased concerns and the need for sources of energy that have low environmental impacts, there is a high probability that many countries will turn to the use of ethanol fuel. Presently, there is a massive use of this type of fuel which necessitates that its production ought to be environmentally sustainable, as well as cost-effective. The implication is that for ethanol to be considered a suitable alternative source of energy its cost of production should be low.
Based on the current projections and the preparation of ethanol on commercial levels, 60% of the cost structure is meant for the acquisition of the necessary feedstocks. The production of ethanol from starch and sugarcane can be relatively cheap. The amount of ethanol produced from corn is remarkably high in comparison to the case when sugarcane is used. In spite of this, more land is needed to effect such change. In addition, when compared with other feedstocks, it is evident that the lignocellulosic biomass is an efficient feedstock in the process of producing high amount of ethanol. This is attributed to the fact that lignocellulosic material can be obtained at a low cost and there are of wide varieties. In addition, these aspects of cellulose contribute greatly to the introduction of bio-related industries that can be used in offering the necessary support to the production of cellulosic ethanol as well as the development and growth of its market.
Large tracks of land are needed to ensure high yield of ethanol from corn. In addition, the production of corn has numerous adverse effects to the environment such as soil erosion and the reduction in water levels. On the other hand, the energy returns from corn are low with respect to the land requirements.
Recommendations
There is a need for the production and use of a source of energy that is sustainable and cost-effective, and environmentally-friendly. According to empirical evidence, the production of ethanol from feedstocks makes it a renewable source of energy. This is attributable to the fact that cellulosic materials, sugar cane, algae, and corn are locally available. While corn leads to a high energy yield per hectare, sugar as a feedstock in the production of ethanol is effective based on its ability to provide a significant balance in terms of greenhouse gas emissions during the production and use of ethanol. On the other hand, cellulosic materials are cheap to acquire and exist in various forms. This analysis shows that the selective production of ethanol can be cost-effective and sustainable.
On the other hand, the production of ethanol from feedstocks guarantees high energy returns relative to the cost of production. For example, the energy balance of ethanol produced in Brazil from sugar is 9.0, while that of the ethanol produced using American corn is 2.0. The implication is that the use of sugar cane in the production of ethanol ensures that all waste products are used efficiently. Based on this assertion, it would be recommendable to use sugarcane in the production of ethanol instead of corn, since such a process is not only cost-effective but also environmentally friendly since all inputs are used.
Therefore, a lot is needed to increase the production of ethanol from various feedstocks. First, the costs of acquiring the feedstocks should be reduced through the improvement of crop yield, adoption of better cropping systems, as well as ensuring that the crops are resistant to pests. In the case of the fermentation process, the use of high cell density is recommendable to enhance high production of ethanol. On the other hand, a lot of care should be taken during distillation to ensure that the right temperatures are maintained for optimal yields of ethanol.
References
Sánchez, Ó.; Cardona, C. Trends in biotechnological production of fuel ethanol from different feedstocks. Biores. Tech. 2008, 99, 13, 5270-5295.
Bothast, R.J.; Schlicher, M.A. Biotechnological processes for conversion of corn into ethanol. App. Micro. Biotech. 2005, 67, 19–25.
Ghosh, P.; Ghose, T.K. Bioethanol in India: recent past and emerging future. Adv. Bioche. Eng. /Biotech. 2003, 85, 1–27.
Macedo, I.C.; Seabra, J.E.A.; Silva, J.E.A.R. Greenhouse gases emissions in the production and use of ethanol from sugarcane: the 2005/2006 averages and prediciton for 2020. J. Biom. Bioener. Biom. 2008, 32,582–595.
Wilkie, A.C.; Riedesel, K.J.; Owens, J.M. Stillage characterization and anaerobic treatment of ethanol stillage from conventional and cellulosic feedstocks. J. Biom. Bioener. 2000, 19, 63–102.
Badger, P.C. Ethanol from cellulose: A general review in: Trends in new crops and new uses; Janick, A., and Whipkey, A., Eds.; ASHS Press: Alexandria, 2002; Vol. 1, pp. 17-21.