Introduction
Opioids fall in a class of drugs that have been used for centuries to manage pain and records show that their usage started in times of ancient Egypt. Research has led to the discovery of special receptors that these drugs work on in the brain and initial studies use morphine to demonstrate a ‘morphine receptor’ (Trescot et al., 2008). A variety of receptors for this class of drugs has since been discovered and new opioids have been discovered and synthetically manufactured.
The pharmacokinetic properties of these newer drugs have generated interest in a number of studies and recently the metabolism of most opioids has been clearly understood. A characteristic of most opioids that has led to their misuse and development of legislation to control their use is the potential addictive properties that they are associated with during their usage. Specific opioids in special dosage forms are also used for the treatment of opioid dependence.
New opioid dosage forms and treatment for opioid dependency have increasingly become part of medical treatments in the recent history with buprenorphine being an important drug for this function (Trescot et al., 2008). This research paper examines the pharmacology of opioids and their use in the treatment of opioid dependency, and specifically it looks at buprenorphine while comparing it with methadone.
Factors affecting oral bioavailability of buprenorphine
The bioavailability of a drug determines the dosage forms and how much of the unchanged form of the drug reaches the systemic circulation (Griessinger et al., 2005). Each route of administration has factors affecting the bioavailability, which are dependent on the body and drug characteristics.
The intravenous route of drug administration provides the highest bioavailability, which is set at 100%. On the other hand, oral administration and bioavailability are affected by the drug characteristics such as solubility, hydrophilic or lipophilic nature, and the receptors that a drug uses to get into the bloodstream (Griessinger et al., 2005).
Incomplete absorption of a drug while in the gastrointestinal system causes low bioavailability. Very hydrophilic drugs are unable to cross the lipid cell membrane, while highly lipophilic drugs are unable to cross the water layer that covers and surrounds the cells involved in absorption (Griessinger et al., 2005).
These elements are some of the factors affecting the oral bioavailability of buprenorphine. The receptor at the absorption site that the drug interacts with is also a significant determinant of the availability of the drug. Some receptors are involved in pumping the drug back to the gut lumen and when these are inhibited, the bioavailability increases (Griessinger et al., 2005).
The oral availability of buprenorphine is poor and it is affected by all the factors described above. However, the most important factor affecting the oral bioavailability of buprenorphine is the high metabolism by the liver and the intestines (Murphy et al., 2013). When buprenorphine is administered orally, the drug is metabolized in the liver and the gut by special enzymes, which leads to the reduction in the proportion of the drug that is available in the bloodstream.
The breakdown of a drug by gut enzymes and processes in the liver has conveniently been referred to as the first pass effect (Griessinger et al., 2005). The main enzyme system responsible for the degradation of the drugs in the liver is the cytochrome P450 (CYP), which is also responsible for the breakdown of buprenorphine. Murphy et al., (2013) posit, “Buprenorphine is converted in the liver primarily by cytochrome P450 (CYP) 3A4 to an active metabolite (nor-buprenorphine with weak intrinsic activity” (p. 316).
The breakdown of buprenorphine by the hepatic enzymes causes a reduction in the bioavailability, hence the efficacy of the drug. One way that can be used to increase the concentration of the drug that reaches the systemic circulation is increasing the dosage that is administered via the oral route. However, an increase in the dosage will mean more side effects as the metabolites affect other receptors.
Therefore, the increase in dosage is not a desirable way of overcoming the first pass effect. Another way in which the first pass effect affects buprenorphine in the liver is the process of glucuronidation that takes place here. According to Murphy et al., (2013), buprenorphine and its metabolite -norbuprenorphine, undergo glucuronidation in the liver and the process reduces its availability.
Various methods have been used to overcome the first pass effect that the drug undergoes when administered through the oral route. Buprenorphine is currently unavailable in oral formulations and the current formulations include the sublingual route and transdermal patches (Murphy et al., 2013).
According to Murphy et al., (2013), methods that can be used to reduce the drug first pass effect include the use of sublingual route, the transdermal routes, and the rectal suppositories. When drugs are administered through the oral route, the absorbed drug and its metabolites are absorbed to the portal system where the drug is taken to the liver and further transformation takes place.
The use of the sublingual route of administration avoids the portal system that takes blood to the liver, and the CYP enzymes break down less of the drug. Once the sublingual formulation is administered, it goes directly to the systemic circulation, hence avoiding the first pass effect (Murphy et al., 2013). According to Trescot et al. (2008), buprenorphine has high lipid solubility and due to this characteristic, the sublingual bioavailability is high.
This method is one of the ways in which researchers and pharmacists have been in a position to avoid the first pass effect. The use of transdermal patches is also common outside the US (Murphy et al., 2013). Transdermal patches are also effective in avoiding the portal system, hence reducing the first pass effect associated with liver metabolism by the Cytochrome P450 system (Murphy et al., 2013).
Drug-receptor relationships
Drug receptors are specific to a certain class of drugs and they are located in areas where the drug will have the desired effect. Opioid receptors have been located in the brain tissue and in other tissues in the body, and specific opioids have specific receptors that they bind.
The main opioid receptors are the “Mu (μ) receptor, the Kappa (κ) receptor (agonist ketocyclazocine), the Delta (δ) receptor (agonist delta-alanine-delta-leucine-enkephalin), and the Sigma (σ) receptors (agonist N-allylnormetazocine) (Trescot et al. 2008, p. 135). Different types of opioids exhibit the several drug-receptor relationships with different receptors being involved.
Opioids can be classified based on their drug-receptor relationships and this classification consists “agonists, antagonists, and partial agonists or antagonists” (Trescot et al., 2008, p. 134).
The affinity of these drugs at their respective receptors can be described as being the “strength of interaction between the drug and its receptor” (Trescot et al., 2008, p. 133). The efficacy of a drug is also considered when discussing the drug affinity and affinity can be described as “the strength of activity of a drug due to its drug-receptor interaction” (Trescot et al., 2008, p. 133).
According to Trescot et al. (2008), an agonist at a receptor is a drug that processes both affinity for the receptor and as a result, it has efficacy. On the other hand, an antagonist has affinity, but its efficacy is absent (Trescot et al., 2008). Drugs with affinity and partial efficacy can be described as being partial agonists at the particular receptor (Trescot et al., 2008).
Partial agonist: – Buprenorphine
Buprenorphine is classified as one of the opioids with low efficacy despite its high affinity, and thus it is a partial agonist (Trescot et al., 2008). This particular drug exhibits affinity at the Mu receptor where it binds by exhibiting its partial effect. It also possesses kappa receptor antagonism, which forms the basis of its use as an analgesic (Trescot et al., 2008).
The drug-receptor activity for this drug has also allowed its use in the management of opioid intoxication, as an abuse deterrent and a maintenance therapy for the detoxification and management of pain (Trescot et al., 2008). Its partial antagonistic effect at the kappa receptor is restricted to a certain level. This ceiling effect means that the drug is only useful up to a certain level and beyond this point, there is limited effect, but just the side effects that can be attributed to any other opioid (Trescot et al., 2008).
Partial agonist: – Oxycodone
Oxycodone is a partial kappa opioid receptor agonist (Murphy et al., 2013). This drug acts on the kappa receptor in different parts of the body to exert its effect and cause the desired effects.
Oxycodone is partial kappa opioid receptor and this aspect means that the drug is not as efficacious as other agonists, and thus it is available in combination form with other related drugs and classes of drugs to provide better efficacy (Murphy et al., 2013). The drugs that are combined with oxycodone to provide a better effect include acetaminophen and aspirin, which act in concert to produce pain relief (Murphy et al., 2013).
Opioid Antagonists: – Naloxone
Naloxone is “an opioid receptor antagonist and it acts at different opioid receptors, thus causing competitive antagonism” (Trescot et al., 2008, p. 139). Naloxone has a competitive antagonism at the delta, Mu, and kappa receptors and according to Trescot et al. (2008), this opioid antagonist has a “high affinity for the mu receptor, but it lacks any mu receptor efficacy” (p. 140).
The drug exerts its effects on both the central nervous system and any other peripheral tissue in the body and the pharmacologic uses are dependent on these actions on the receptors.
Naloxone is mainly used to reverse adverse opioid effects and it is useful in the maintenance of other treatments, deterrence to the use of opioids, and detoxification (Trescot et al., 2008). However, its main use is in the management of opioid toxicity due to its antagonistic nature at the opioid receptors.
Combination of the drug with other drugs such as buprenorphine is also used to prevent users from abusing the second drug when given intravenously (Trescot et al., 2008). Studies are also being conducted to establish whether the drug can be used to suppress tolerance to other drugs such as oxycodone (Trescot et al., 2008).
Opioid Antagonists: – Naltrexone
Naltrexone is also an opioid receptor antagonist and like naloxone, it acts at different opioid receptors by causing competitive antagonism (Trescot et al., 2008). Naltrexone also exhibits competitive antagonism at the delta, Mu, and kappa receptors and Trescot et al. (2008) state that like Naloxone, this opioid antagonist has a “high affinity for the mu receptor, it lacks any mu receptor efficacy” (p. 140).
The drug also exerts its effects on both the peripheral and central nervous system and many other tissues in the body with the receptors. The pharmacologic uses are similar to those of Naloxone and they are dependent on these actions on the receptors.
Naltrexone is mainly used to reverse adverse opioid effects and it is useful in the maintenance of other treatments, deterrence to the use of opioids and in detoxification (Trescot et al., 2008). Like Naloxone, the main use of Naltrexone is in the management of opioid toxicity due to its antagonistic nature at the opioid receptors.
Combination of the drug with other drugs such as buprenorphine is also done to prevent users from abusing the second drug when given through the intravenous route (Trescot et al., 2008). Studies are also being conducted on Naltrexone as with Naloxone to establish whether the drug can be used to suppress tolerance to other drugs such as oxycodone (Trescot et al., 2008).
Opioid Agonists-Antagonists: – Pentazocine
Opioid agonist-antagonists have poor efficacy at the mu receptor, but they have agonistic properties at the kappa receptors (Trescot et al., 2008). Pentazocine is a good example of partial agonist-antagonists and it exhibits all the characteristics that the group processes.
Partial agonist-antagonism means that the affinity of Pentazocine at the mu receptors is high with a corresponding low efficacy at the same receptor (Trescot et al., 2008). The drug is used as an analgesic like many opioids, but it also has a ceiling effect where increase in dosage will not lead to increase in analgesic properties, but only the toxicity of the drug (Trescot et al., 2008).
Respiratory depression
Reparatory depression is a major side effect property of opioids and it is exhibited by the reduction in the respiratory rate leading to the accumulation of carbon dioxide in the blood stream. Opioid activity on the mu receptors is the main known cause of respiratory depression in individuals using opioid drugs.
This side effect is potentially fatal and it varies with the tee of opioid that is used. It is also dependent on the personal characteristics of individuals and it is more pronounced in children as compared to adults. Buprenorphine and morphine are some of the opioids that exhibit this property of respiratory depression. However, these two opioids have different characteristics in their respiratory depression as discussed later in this paper.
Difference between buprenorphine and morphine
Morphine causes direct respiratory depression by acting on the mu receptors in the brain (Trescot et al., 2008). The drug binds to the receptor in the nucleus accumbens in the brain and the result of this activity is a decrease in response to carbon dioxide concentration rise in the bloodstream (Trescot et al., 2008).
When the reaction to high levels of carbon dioxide in blood is inhibited, the respiratory level decreases, which lead to the retention of more carbon dioxide in the blood. The result of accumulation of carbon dioxide in blood is a drop in the pH, thus resulting in respiratory acidosis. According to Trescot et al. (2008), the response curve shifts towards the right.
One known characteristic of morphine is that acidosis leads to increased delivery of the drug to the brain tissues (Trescot et al., 2008). As the blood carbon dioxide increases due to the depression of the nucleus accumbens and respiratory acidosis occurs, the amount of morphine that is delivered to the brain increases and this aspect further acts to cause profound respiratory depression that may be fatal (Trescot et al., 2008). Therefore, increase in morphine causes an increase in the respiratory depression and it can be said to be dose dependent.
Respiratory depression occurring from buprenorphine is not central, as it is evident with morphine. On the contrary, buprenorphine induces respiratory depression by acting on the mu receptors located in the lungs and not in the brain (Ohtani et al., 1997). As stated earlier, buprenorphine is a partial agonist, and thus the respiratory depression that it exhibits has a ceiling effect (Murphy et al., 2013).
The binding of this drug to the mu receptors is also stronger as compared to the binding by morphine, and thus the effects take longer and they may be prolonged (Murphy et al., 2013). The tight binding means that reversal using the opioid antagonists like naltrexone is harder as compared to other opioids.
Importance of the difference
The differences between the two drugs are significant in the application and daily use of the drugs in the management of pain and opioid toxicity. By exerting respiratory depression centrally, morphine can be used in limited doses, which must be monitored.
The toxicity that can come from this drug is also profound and it may be fatal. Higher doses of buprenorphine can be used as compared to the safest doses of morphine. Another implication is that the management of respiratory depression for buprenorphine may be hard using opioid antagonists such as naloxone as compared to the respiratory depression that is caused by morphine.
Dose-response curves
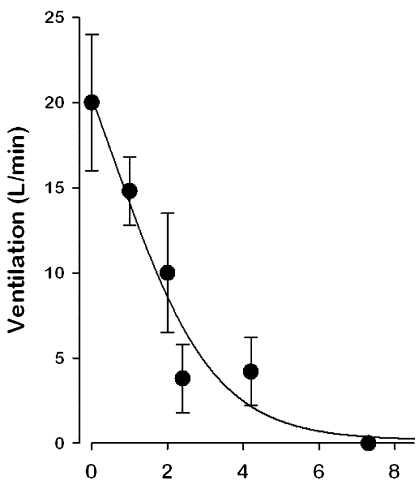
The dose response curves for the respiratory depression caused by morphine and buprenorphine are different. The respiratory depression in the curves can be represented by the decrease in ventilation rated after administration of the two drugs.
Graph showing the ventilation rate against the concentration of buprenorphine (source: Ohtani et al., 1997).
Transdermal dosage form of buprenorphine
Buprenorphine is available in the oral and transdermal route and a patch is applied to deliver constant drug doses for a period. The transdermal application of buprenorphine has several differences in the dosage, bioavailability, and efficacy to the use of the oral route.
The transdermal route is better tolerated compared to the oral route with fewer side effects being experienced due to the use of this path of drug administration (Pergolizzi et al., 2010). The common side effects at the area of application are the development of erythema and pruritus (Pergolizzi et al., 2010).
The transdermal route of application of methadone also lasts longer as compared to the use of the oral preparation. According to Pergolizzi et al., (2010), the patch acts as a depot for the drug, thus ensuring a constant supply of the same on the body’s demand.
The oral route requires frequent administration of the drug when used to manage pain and in the management of opioid toxicity. On the other hand, a transdermal patch has been used for over three days with researchers showing that it is still efficacious after the third day (Pergolizzi et al., 2010).
As earlier described, the first pass effect of a drug is important in the determination of its bioavailability. Through the application of buprenorphine orally, there is a considerable chance of the drug going through the first pass effect, which results in the reduction of the drug’s bioavailability.
The use of the transdermal patch allows the bypass of the liver and the gastrointestinal system, thus reducing the first pass effect (Pergolizzi et al., 2010). However, the transdermal route is a slower method of delivery of buprenorphine and it cannot be used to deliver the drug when it is needed more urgently. On the other hand, the sublingual and oral route, according to Pergolizzi et al. (2010), is a faster way of delivery of the drug.
Buprenorphine vs. methadone
Methadone is a synthetic diphenylheptane and it acts on the mu opioid receptor as an agonist (Trescot et al., 2008). This drug is unique with properties different from those exhibited by other opioids. The difference with other opioids will be compared in this section by using buprenorphine. The half-life of the two drugs, time of onset, and duration of effect and the dosing regimens will be used in the comparison.
Half-life
Trescot et al. (2008) posits that the half-life of a drug “is the time that it takes for its blood concentration to reduce by half” (p. 144). The plasma half-life of buprenorphine is 3-5 hours, which means that the drug plasma concentration reduces by half after 3 to 5 hours of administration.
The faster reduction in concentration of the drug when given through the sublingual route means frequent administration and this aspect underscores a setback in the management of intoxication since a more frequent dosage is required (Trescot et al., 2008). The transdermal patch also has the same half-life, but the availability is high and it provides a depot for the drug.
Methadone has a long half-life as compared to buprenorphine and this aspect can be attributed to the high lipid solubility (Trescot et al., 2008). High lipid solubility means that the drug is widely distributed in tissues, especially in fat tissue, which provides methadone with a very long elimination phase (Trescot et al., 2008).
Due to this aspect, the half-life is stated to be between 12 and 150 hours, and hence methadone may be administered once daily or longer than buprenorphine. A comparison of the two drugs shows that methadone has a longer half-life as compared to buprenorphine.
Time of onset/duration of effect
The time of onset of effects of a drug is the time that passes between the administration of a drug and the patient to experience its effects. In the case of buprenorphine, the onset of effects is three to four hours after administration through the sublingual route (Murphy et al., 2013).
This onset of action is slower as compared to other opioids and it is desired where the treatment of toxicity of opioids is to be treated (Murphy et al., 2013). The onset of action for buprenorphine, while using the transdermal route, is also slower as compared to the sublingual route. The duration of action of this drug is also long and it lasts for 12-150 hours
Methadone is similar to buprenorphine in the onset of action, and the process is even slower when given orally. However, the take taken for methadone to act is longer than that of buprenorphine and this case has been reported in some literature to be 4-5 days. This observation means that methadone can be administered in longer doses duration such as once daily and its effects can last longer (Murphy et al., 2013).
Dosing regimens used
The dosing regimen can be described as the formulation, route of administration, the dosage, and interval of administration of a drug (Murphy et al., 2013). Buprenorphine is available as a sublingual tablet, while methadone is available in most places as an oral liquid formulation (Murphy et al., 2013).
The onset of action for buprenorphine, as stated earlier, is slow and this characteristic is compatible with the slow onset of action that methadone has in its functioning. The buprenorphine tablets are dissolved under the tongue, while the liquid formulation of methadone is swallowed. Alternate day dosing for buprenorphine is also possible, while it is impossible for methadone.
Conclusion
Opioids have been used in the management of pain for a long time and a number of factors have contributed to their preference over other classes of drugs. This paper has described some of the receptors that opioids bind to exert their effects and the receptor-drug interaction has been described by giving examples. A number of opioids are also used in the management of opioid toxicity based on the receptor interactions that they display.
The paper focused on buprenorphine to examine some of the pharmacokinetic properties of opioids. Respiratory depression was stated as a major side effect of the opioids and there are established differences between respiratory depression provided by buprenorphine and that produced by morphine. A comparison between methadone and buprenorphine was also provided in the paper.
References
Griessinger, N., Sittl, R., & Likar, R. (2005). Transdermal buprenorphine in clinical practice-a post-marketing surveillance study in 13,179 patients. Current Medical Research Opinion, 21(1), 1147–1156.
Murphy, L., Fishman, P., McPherson, S., Dyck, D., & Roll, J. (2013). Determinants of buprenorphine treatment for opioid dependence. Journal of Substance Abuse Treatment 46(3), 315-319.
Ohtani, M., Kotaki, H., Nishitateno, K., Sawada, Y., & Iga, T. (1997). Kinetics of respiratory depression in rats induced by buprenorphine and its metabolite, norbuprenorphine. Journal of Pharmacology and Experimental Therapies, 281(1), 428–433.
Pergolizzi, J., Aloisi, A., Dahan, A., Filitz, J., Langford, R., Likar, R., Mercadante, S., Morlion, B., Raffa, R., Sabatowski, R., Sacerdote, P., Torres, L., & Weinbroum, A. (2010). Current Knowledge of Buprenorphine and Its Unique Pharmacological Profile. Pain Practice, 10(5), 428–450.
Trescot, A., Datta, S., Lee, M., & Hansen, H. (2008). Opioid Pharmacology. Pain Physician, 12(2), 133-153.