Introduction
Refrigeration is a vital cooling process essential to various industries and everyday life. Energy efficiency can therefore be attained when a person understands the coefficient of performance for the refrigeration cycle. The cooling performance requires regulation to ensure that the energy use is optimized. Mechanical refrigeration, also known as the vapor-compression cycle, is one of the most widely used methods for determining cooling efficiency (Li & Yu, 2019).
A refrigerator is a reverse heat pump that essentially takes heat from a body and cools it. According to the second law of thermodynamics, the reverse movements of heat can only be facilitated by an external force. A basic refrigeration cycle, therefore, focuses on four key processes: expansion, evaporation, condensation, and compression.
Theory
The refrigeration cycle draws partly on the second law of thermodynamics and the four key processes involved in cooling. The processes take place at distinct layers of refrigeration. The primary role of the compressor is to increase the temperature and pressure of the refrigerant vapor.
The condenser transfers heat from the refrigerant to the surrounding air, causing it to condense. The expansion occurs at a valve that controls the flow of refrigerant and ensures a drop in its temperature (Li & Yu, 2019). The evaporator is the final stage of the refrigeration cycle, where the refrigerant is converted to a vapor. The cooling process occurs through four repeating cycles.
Each of the four refrigerator sections has a distinct equation that determines the outcome.
Evaporator:
Condenser:
Compressor:
It is imperative to note that the compressor has an exceptional isotropic efficiency, which is given as shown in the equation below:
The pressure ratio
Coefficient of performance, commonly referred to as COP
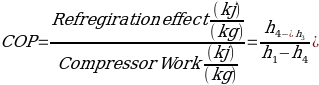
Aim and Objectives
The primary aim of this experiment was to determine the coefficient of performance of a refrigerator and to plot the vapor-compression process on a pressure-enthalpy diagram. Furthermore, the experiment examines the impact of pressure and temperature changes on the sub-valves.
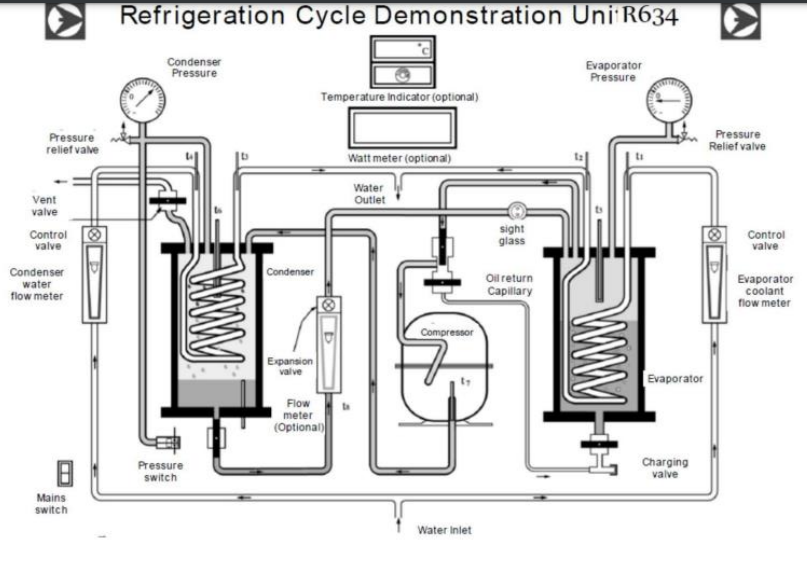
Apparatus and Experimental Setup
Figure 1 illustrates the apparatus and experimental setup. Figure 2 illustrates the experimental arrangement. The central apparatus for the investigations included a pressure gauge, evaporator, expansion valve, thermometer gauge, compressor, condenser, and piping equipment. Water was used as one of the primary fluids in the refrigeration process.

Experiment Procedure
The sub-cooler of the refrigeration system was put to an off state, and the throttle was put to the capillary. Once the off-state was confirmed, the compressor was put in an on-state and allowed to run for about fifteen to twenty minutes to determine the system’s efficiency. Once the first readings were taken, the throttle was set to a new level (TXV), and a second set of readings was taken. The sub-cooler was turned on again, and the process was repeated.
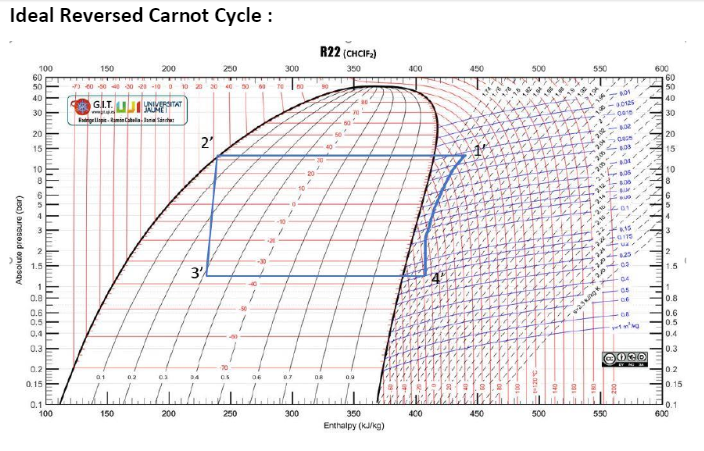
The readings taken from the experiment were recorded in Table 1 below. In addition to the recorded temperature and pressure values, the corresponding enthalpy values were obtained from a p-h chart. Furthermore, the results were plotted on a pH plot and then compared with the ideal Carnot cycle.
Measurements and Results
Table 1 – Results and different tests
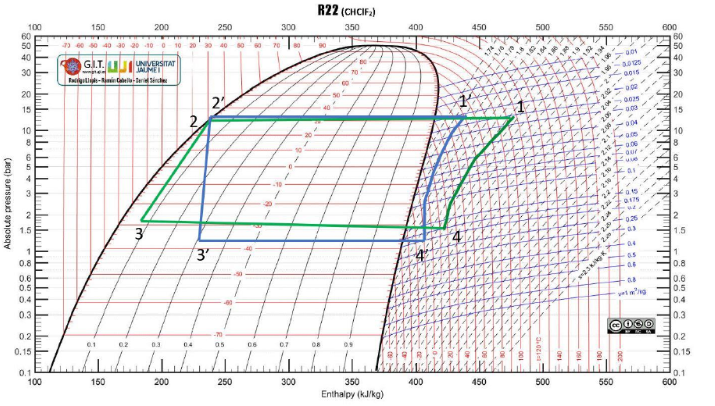
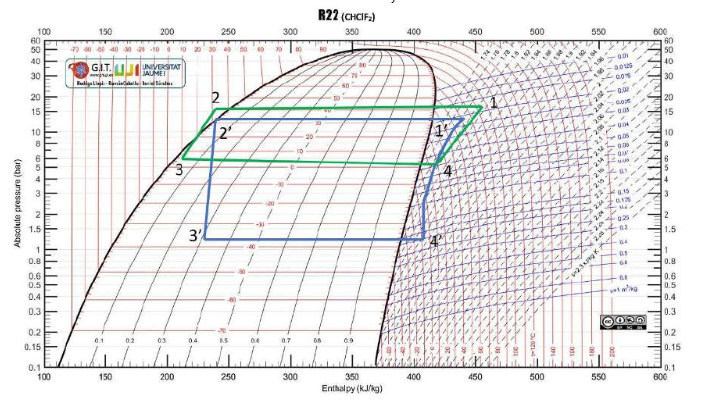
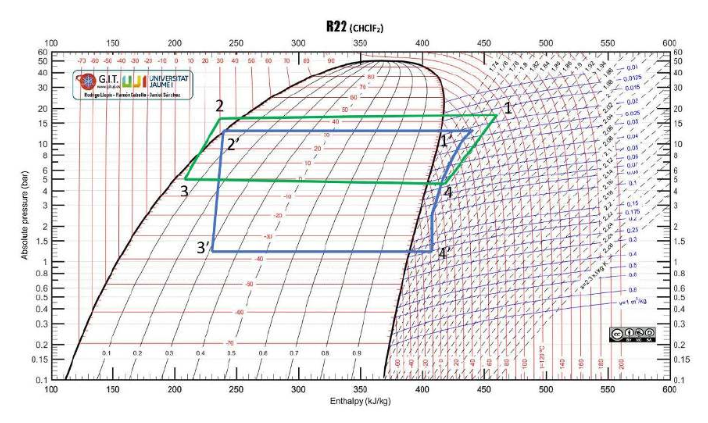
The ideal car note cycle was used to compare the different tests to understand the refrigeration process. The ideal cycle is shown in Figure 3 below, while Figures 4, 5, 6, and 7 are the four different tests conducted.

Analysis
Four main processes are involved in completing the experiment. The first step is compression, in which the refrigerant remains in its gaseous state. It enters the refrigeration chamber at low pressure and temperature. As shown in the curves, the compression chamber reduces volume and increases pressure, thereby increasing temperature.
Condensation is the second step, in which the gas is cooled with water while being kept at constant pressure. The third step involves expansion and throttling, which cool the hot gas by decreasing pressure (Li & Yu, 2019). The final step is the evaporation phase, during which the refrigerant is converted into a gas as it absorbs heat from the fridge’s contents. Consequently, the contents remain unconscious as the refrigerator’s efficiency remains higher.
Discussion
The real-world data were lower than the values of the ideal cycle, as the assumptions made were based on a theoretical cycle. Some factors, such as constant pressure, are impossible in the real world, and atmospheric conditions fluctuate (Li & Yu, 2019). It is prudent to note that even the room temperature and pressure are not constant and fluctuate from one value to another.
The difference between the ideal Carnot and the real-world tests was lowest at the third test point. Since the third test, which employed sub-cooling, proved to be the most effective, optimal refrigeration conditions could be achieved by appending the capillary to a subcooling system. Sub-cooling is crucial in refrigeration because the vapors remaining after the initial cooling can compromise the process (Li & Yu, 2019). The subsystem ensures that only liquid passes through and reaches the throttling device.
Conclusion
The lab was a particular exercise that helped me understand the refrigeration process. Besides the working principle, the lab helped to understand the impacts of varying pressure and temperature when using sub-coolers and throttling devices. A sub-cooler was used to determine the coefficient of performance. The curves obtained were compared with the ideal Carnot cycle to identify methods that improve optimal performance. It was concluded that a capillary tube and a sub-cooler system were the key components to achieve optimal performance.
Reference
Li, Y., & Yu, J. (2019). Thermodynamic analysis of a modified ejector-expansion refrigeration cycle with hot vapor bypass. Journal of Thermal Science, 28, 695-704.