Aims and objectives
The two laboratory practicals aimed at determining whether or not patients’ serum samples contained antibodies that could react against gastric proton pump through the application of Western blotting and immune-histochemistry tests. In addition, the practicals aimed at suggesting determining the category of patients who had pernicious anemia.
Introduction
Pernicious anemia is the last presentation of chronic gastritis, which is an autoimmune disease that affects the stomach. Pernicious anemia is characterized by swelling of the stomach, overproduction of autoantibodies such as intrinsic factor and proton pump. All these proteins are secreted by the parietal cells of the stomach (Toh, van Driel & Gleeson 1997). Research has also demonstrated that gastric atrophy is characterized by diminishing the mass of the stomach (gastric atrophy). The pathophysiology of the disease has been correlated with the activity of autoantibodies, which attack intrinsic factor that is essential for the absorption of vitamin B12 (Toh et al 1997). In order for the erythrocytes to mature, they require the activity of vitamin B12. The absorption of the vitamin is facilitated by an intrinsic factor that is within the stomach. Thus, when the activity of the intrinsic factor is compromised, the amount of vitamin B12 absorbed is greatly reduced. Hence, anemia occurs. Gastric glands contain parietal cells, which are the main sources of hydrochloric acid in the stomach. The acid is an important agent in the initial stages of digestion. However, it is only secreted when the activity of H+/K+-ATPase (Toh et al 1997). This enzyme is a proton pump that has been shown to be involved in essential transport pathways within cells. In addition, the enzyme has unique beta and alpha subunits. The subunits react to the autoantibodies in the serum of human beings. Particularly, if a patient is suffering from pernicious anemia, then his or her serum reacts alpha or beta subunits of the proton pump. Thus, the diagnostic kit that is used to test for the presence of pernicious anemia involves the use of the alpha and beta subunits (Toh et al 1997).
Methods
This report is written to reflect the activities that were associated with practical two and practical three with regard to pernicious anemia. Practical two involved the separation of proteins that were isolated from mouse stomachs through the application of SDS-PAGE. Practical three involved the performance of Western blotting with regard to the proteins separated in practical two. In addition, practical three was characterized by the staining of a mouse stomach section with hematoxylin and eosin in order to make calls within the stomach more visible under the microscope.
SDS-PAGE and Western blotting
SDS-PAGE is a commonly used method in the study of components of protein mixtures. In this method, proteins are dissolved in sodium dudecyl-sulfate (SDS), which binds to the portions of proteins that are hydrophobic. Once SDS accesses the hydrophobic regions of proteins being studied, it makes them unfold and become linearised. Unfolded proteins achieve an overall negative charge. Disulfide bonds within proteins are broken through the use of reducing agents such as 2-beta mercaptoethanol. When such proteins are run on gel electrophoresis, they migrate from the negative side of the equipment to towards the positive portion, where they could combine with the opposite charges at the anode. In order to identify the identity of the isolated proteins, Western blotting is carried out. This method involved the transfer of proteins separated through the SDS-PAGE onto a nitrocellulose membrane. Afterward, probing was conducted using antibodies (Frost, Sparks & Grizzle 2000).
The nitrocellulose membrane was retrieved from the storage pouch and the remaining membrane was divided into the number of patients being tested. About 100 µl of the sera was added onto each strip of the membrane and the membrane portions were incubated at room temperature for 1 hour. The strips were then washed in TBST and rocked. The strips were placed onto a parafilm. About 100 µl of the secondary antibody was distributed onto the films. The strips were washed in TBS with rocking. The membrane was placed into a Chemidoc apparatus that could aid the detection of the substrate. Two images were obtained for interpretation.
Electrophoresis
A precast polyacrylamide gel was placed in an electrophoresis tank, which was filled with a running buffer that could support the flow of current that could move the proteins being studied. In order to prepare a stomach protein sample isolated from the stomach of a mouse, 50µl of 5x SDS buffer was added to a tube that contained 200 µl of stomach protein. The tube contents were thoroughly mixed to obtain a homogenous solution. The tube was sealed and kept at room temperature. A molecular weight marker was added to the first well of the gel. The marker was used so that it could give references to the distances traveled by protein samples. Each well was loaded with 25 µl of the protein samples. The electrophoresis was run for 1 hour. The next step involved the transfer of proteins in the gel into a nitrocellulose membrane. Once the transfer was completed, the membrane was placed into a vessel that contained 50mL 0.1% Ponceau stain and placed on the rocker for 1 minute. The membrane was rinsed through the use of deionized water until the protein bands were visualized. The membrane was labeled using a pencil so that the correct interpretations regarding the source of proteins could be made. Afterward, the membrane was rinsed using 50mL 0.1M NaOH. This was done to clear the stain on the nitrocellulose membrane. It was also rinsed in 50mL TBS. Nonspecific binding of proteins was prevented from occurring by the addition of 50mL TBS, which was mixed with 5% skim milk powder. The membrane was clearly labeled and stored in a moist environment.
Morphology of the stomach
The slide containing the cells from the stomach of a mouse was fixed in ethanol for 2m minutes. Rehydration was done in running tap water for about 30 minutes. The slide was incubated in hematoxylin solution for 2 minutes, rinsed in water and shortly dipped in alcohol that contained acid. To avoid the contents of the slide from being exposed to the prolonged effects of the acid, the slide was immediately rinsed in water. Afterward, the slide was washed in eosin Y for 4 minutes and in 90% ethanol for 30 seconds, and absolute ethanol for 30 seconds. The slide was air-dried and observed under the microscope using different magnifications. The diagrams were labeled for easy identification.
Immune-peroxidase staining of mouse stomach sections
Immuno-histochemistry technique is utilized to detect the presence of particular proteins (antigens) in the tissue section. In addition, the method is applied in detecting antibodies in a serum sample. In the practical, the presence of anti-proton pump antibodies was assessed using the serum. In that case, the sections of the mouse stomach contained the proton pump antigen. Once unbound serum was washed away, the section was incubated with a secondary antibody that was very specific for human immunoglobulin molecules. The secondary antibody was conjugated to protein (an enzyme), which could convert the substrate to a colored product that could be easily recognized through the reading apparatus. A pair of two students performed the experiment, which was based on two slides that contained the same serum samples that were tested through Western blotting.
The procedures were as follows: The slide containing the mouse sections was incubated in xylene for 2 minutes and in ethanol for 2 minutes. The slide was rinsed in tap water to remove the washing reagents. The endogenous peroxidase activity that could negatively impact the quality of the results was removed by washing the slide in 0.3% hydrogen peroxide and PBS for 30 minutes. The slide was washed in PBS buffer for 2 minutes and incubated in 1% BSA and PBS for 30 minutes. The procedure was done to block the non-specific binding of proteins. However, the slide was not washed after the procedure was completed. The blocking solution was removed and 100 µl patient’s serum was added to the section and incubated for 20 minutes. Afterward, the slide was washed for 6 minutes in PBS. Sheep anti-human (50 µl) anti-immunoglobulin solution was added to the section and incubated for 45 minutes. The slide was washed in PBS for 6 minutes. The next step involved the addition of 100 µl of Dab substrate to the section and incubation was done for 10 minutes. In order to remove the washing buffers, the slide was washed by dipping into hematoxylin for only 3 seconds and in water for 30 seconds. The slide was washed in ethanol for 2 minutes. A small drop of DPX mounting medium on the section and covered with a coverslip. However, the excess mounting medium was cleared from the slide through the application of clean cotton material. At this stage, it was determined whether or not the parietal cells had been stained. A diagram was prepared based on a stomach section that had a positive antibody reaction. The diagram was labeled appropriately for easy interpretation.
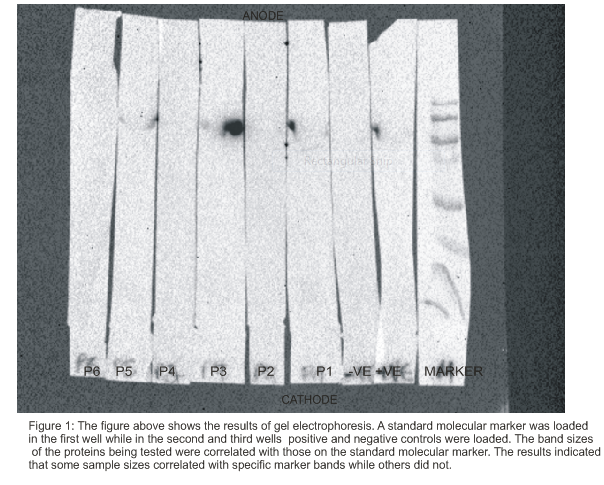
SDS-PAGE
The results that were obtained with regard to SDS-PAGE are shown in figure 1. The results illustrate the molecular sizes of the various protein components that were in the samples being tested. The aim of the SDS-PAGE was to show whether or not there was the presence of the autoantibodies that are associated with pernicious anemia (Frost et al 2000). The expected size of the protein that was being detected by the sera from pernicious anemia patients was 65-70kD. Thus, the samples that contained the protein of interest could correlate with the molecular sizes of the marker at 65-70kD. The size of the protein is commonly associated with the beta subunit of the proton pump in gastric cells that play great roles in the absorption of vitamin B12 (Pérez-Pérez 1997). The samples that showed the presence of the protein sizes, then it indicated that the patients were suffering from pernicious anemia. On the other hand, if patients did not have the serum proteins of the specific sizes that correlate with the condition, then it implied that they did not suffer from the disease. Another method that could be used to detect and quantify the presence of serum proteins with 65-70kD could be the ELISA test. This could be based on the antigen-antibody reactions. The presence of the antibodies could be detected through the identification of some colors of reaction products while the amount of the antibodies could be known by determining the intensity of the colors of the products formed.
Immunochemistry
Figure 2 shows the results that were obtained from immunoblotting. The first image in the figure displays the image of the standard marker while the second image shows the image blot with regard to the samples tested. Specific colors were given out by the products that were formed when the molecules in the nitrocellulose membrane reacted with enzyme conjugates. Particularly, certain colors could indicate the presence of the reactions, which were characterized by antibody-antigen complex formations. The sheep anti-human Ig-HRP conjugate that was used in the Western and immuno-histochemistry was specific to the proton pump antigen. The antibody was raised in a transgenic mouse to increase its specificity for the antigen. The enzyme that was conjugated to the antibody was ubiquitin ligase.

Figure 3 provides a table that summarises the results of microscopy with regard to immune-staining of mouse stomach sections. The table indicates the reaction results of the six samples, positive control and negative control. All the controls and samples were done in two sets. The negative control gave the most consistent results because all the reactions produced negative results. The summary of the results shows that P1 and P2 samples were positive and negative respectively. The results of the other samples could not be relied upon because the third reaction in all the samples did not produce results.
Figure 3: A table showing the summary of sample results with regard to gel electrophoresis.
Conclusion
The results are quite significant in the context of the diagnosis of pernicious anemia. They show that the proton pump that is associated with gastric cells is largely involved in the pathophysiology of pernicious anemia. All the aims of the practical were achieved and the results provide a better understanding of the disease that was studied. However, the results that were obtained on the mouse stomach sections show that there was contamination of the samples. Thus, sterility in the laboratory was not achieved. In addition, group members could have mixed some samples and reagents unknowingly. The problem could be avoided in the future through the implementation of better approaches in the context of sterility and labeling of reagents and samples. The problem that was experienced in the low quality of the images could also be avoided. The experiment could be improved by using standardized volumes of reactions, which could improve the quality of results (Bonini, Plebani, Ceriotti & Rubboli 2002; Kuperman et al 1999). This could require the determination of the right volumes of samples and reagents before conducting the real experiment.
References
Bonini, P, Plebani, M, Ceriotti, F, & Rubboli, F, 2002, ‘Errors in laboratory medicine’, Clinical chemistry, vol. 48, 5, pp. 691-698.
Frost, AR, Sparks, D, & Grizzle, WE, 2000, ‘Methods of antigen recovery vary in their usefulness in unmasking specific antigens in immunohistochemistry’, Applied Immunohistochemistry & Molecular Morphology, vol. 8, 3, pp. 236-243.
Kuperman, GJ, Jonathan, M, Tanasijevic, MJ., Ma’Luf, N, Rittenberg, E, Jha, A,… & Bates, DW, 1999, ‘Improving Response to Critical Laboratory Results with Automation Results of a Randomized Controlled Trial’, Journal of the American Medical Informatics Association, vol. 6, no. 6, pp. 512-522.
Pérez-Pérez, GI,1997, ‘Role of Helicobacter pylori infection in the development of pernicious anemia’, Clinical infectious diseases, vol. 25, 5, pp. 1020-1022.
Toh, BH, van Driel, IR, & Gleeson, PA, 1997, ‘Pernicious anemia’, New England Journal of Medicine, vol. 337, no. 20, pp. 1441-1448.