Summary
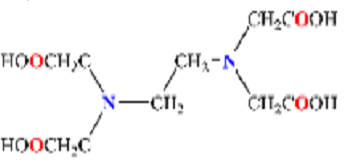
Metal ions in solution can be determined by titration with complex forming ions. EDTA is one of the most popular complexing agents that is suitable for this purpose because it complexes with multiple metal ions. EDTA forms stable complexes with many metals anions. The bonding is through the carboxyl oxygens and the two nitrogens.
Introduction
Titrometry is one of the techniques that chemical analysts use to determine the concentration of metal ions in mineral water. A complexing agent is necessary to facilitate accurate estimation of metal ions concentration in water. EDTA is the most popular complexing agent scientists use in the titrometry of metal anions in mineral water.
Complexes form from the interaction of metal ions with tetrabasic ions from an acid. The nickel chemical structure includes an oxygen atom from a molecule of water that substitutes an oxygen atom from EDTA at the sixth position of the structure. This chemical reaction can be represented in the equation below (Gordon & Lim 2012):
Mn+ + Y4- ↔ MYn-4
Y4- represents the tetrabasic anion of EDTA.
Titration using EDTA involves indicators that are specific or selective. These indicators form complexes with dissolved metal ions, but EDTA complexes with metal anions preferentially, liberating the indicator at the stoichiometric point. Both the complex and the indicator are colored. Thus, the liberation of indicator causes a color change that is unique to the metal anion (Gordon & Lim 2012). A buffer is necessary to provide suitable pH for the complexing of EDTA with metal. Masking agents may be added to the mixture in the flask to avert interference from extraneous metal anions.
Method
A 0.01 M EDTA solution was prepared for the titrometry. 25.00ml of aliquot was measured using a pipette into the Erlenmeyer flask. A 2 mL of ammonia, buffered at pH 10, were added into the Erlenmeyer flask before adding 0.2 g of ascorbic acid, and a few drops of Eriochrome Black T Indicator. The water sample with Eriochrome Black T indicator was titrated with the standard solution of EDTA until the color of the mixture in the flask turned from wine red to clear blue. The addition of titrant was done slowly because the change of color towards the endpoint is usually steady. The titration was repeated thrice to obtain three concordant results.
Results
- First reading of titration = 17.5
- Second reading of titration = 17.4
- Third reading of titration = 17.5
Concentration of Mg2+ = 52.0
- M (EDTA) = 0.01 M
- V (EDTA) = 17.18
- n = C*V
- n = 0.0100*00175 = 0.175
Calculations


Discussion
Since Eriochrome Black T acts as an acid-base indicator, the titration should be performed at a stable pH. Eriochrome Black T can occur in three different forms as shown below depending on the pH of the environment.
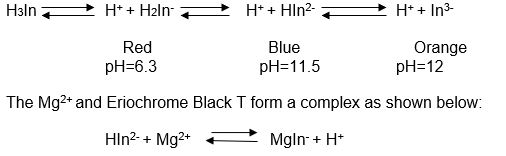
This complex is wine red in colour and less steady compared to Mg (EDTA)2 complex. In the event of titration of a solution containing Mg ions, the EDTA complexes with free Mg2+ first before reacting with MgIn– complex to liberate the indicator and yield blue color.
These are the results I have expected as there can be more than one type of metal ion in mineral water. I used more EDTA because of extraneous metal ions that the titrant formed complexes with. Thus, more EDTA was necessary to reach the endpoint.
Because the hardness of mineral water differs based on the source, the volume of sample water for every titration ought to be determined following the first experiment with a choice volume of the water sample. An experimenter must dilute the standard 0.01 M EDTA solution for use with extremely soft water. Conversely, little water samples or higher molar EDTA solutions are used for very hard water (Blackman, Bottle, Schmid, Mocerino & Wille 2012). Often, the test water contains sufficient Mg ions to facilitate direct titration.
Ions of aluminum, copper, nickel and iron if they are present, even in negligible concentrations, complex with the indicator obscuring the sharpness of the color change at the endpoint. Consequently, the experimenter should add KCN to eradicate all the extraneous metal ions.
The experimenter cannot titrate Ca2+ by itself to the endpoint of the Eriochrome Black T, because the association constant for a calcium-Eriochrome T complex is negligible. The titration of a metal ion is non-selective. The success of a non-selective titration of a metal ion is usually facilitated by the addition of a determined amount of Mg ions if the association constant of the other metal ion with EDTA exceeds that of Mg2+ with EDTA. In addition, the association constant for the metal-indicator complex must be low for the mentioned situation to take place (Housecroft & Constable 2010).
Among all metal ions for which the titration with EDTA applies, a Magnesium ion is apparently the most insensitive to solutes that are capable of forming complexes with EDTA. Thus, experimenters are able to perform all magnesium titrations with a single indicator.
Conclusion
In the most popular way of titrating magnesium with EDTA (Schwarzenback’s technique), Eriochrome Black T is the most popular indicator of choice. It allows the experimenter to adjust the medium of titration to pH 10 by adding ammonia or ammonium chloride buffer. Adding 0.2 g of ascorbic acid helps remove oxygen from the titrate to avert oxidation of the indicator by air. The color of titrating is red at pH 10 in the presence of Mg2+ before titration, because of magnesium-Eriochrome Black T complex. Thereafter, it turns magenta because magnesium-EDTA complex immediately before the reaction reaches the stoichiometric point. After the endpoint, it turns blue with titration of excess EDTA.
References
Blackman, A, Bottle, S, Schmid, S, Mocerino, M & Willie, U. 2012. Chemistry, John Wiley & Sons Australia, Milton Qld. Chapter 13.
Gordon, J & Lim, K 2012, SLE235 Chemical Systems Laboratory Manual, Deakin University of Australia, School of Life and Environmental Sciences, Faculty of Science & Technology:
Housecroft, C & Constable, E 2010, Chemistry, Prentice Hall. Chapter 23.