Abstract
Even mild muscle weakness can cause children discomfort and reduce their quality of life. Such a severe genetic disorder as Duchenne muscular dystrophy (DMD) leads to tremendous consequences for the muscular system and affects children very early in their development. Understanding the causes and implications of DMD for young children is imperative not only for raising awareness of the problem and recommending solutions for the management of the condition. The purpose of this paper is to provide an in-depth exploration of DMD, including its background and symptomology, genetic significance, diagnostic process and management, as well as prognosis.
Introduction
Duchenne muscular dystrophy (DMD) is a genetic condition that is characterized by the continuous deterioration and weakness of muscle associated with changes in the composition of a protein called dystrophin, which maintains the functioning of muscle cells. The disorder is rare and affects males predominantly, with women being diagnosed with it only in exceptional cases. DMD causes the muscles in the body to become less resistant to physical impact and get damaged over time. Thus, the purpose of the paper is to explore Duchenne muscular dystrophy in great detail, including its causes, demographic data, signs and symptoms, the overall effect on the body, discuss the diagnostic process and prognosis for the disease.
Background and Symptoms
Duchenne’s dystrophy develops as a result of a genetic mutation that does not allow the body to produce dystrophin, a protein needed to build muscles. Without the availability of enough dystrophin in the body, muscle cells weaken and become damaged. Children diagnosed with the disease at the early stages of life experience significant issues with walking and breathing, which decreases the quality of their lives (Birnkrant et al., 2018). Eventually, the muscles responsible for breathing stop working, which causes death. DMD is an irreversible, progressive disease that currently has no cure that could have alleviated the burden of the disease.
The estimated global prevalence of DMD is 4.78 per 100,000 males (Walter & Reilich, 2017). The implications of the condition also include a yearly disease burden of more than $130 million, without including costs for respiratory management, extra direct costs per one patient, as well as additional medical aids (Walter & Reilich, 2017). According to the official website dedicated to DMD, the average age of diagnosis is 5 years, while it takes 2.5 tears between the initial symptoms and diagnosis (“Duchenne muscular dystrophy: The basics,” 2019).
Being one of the most severe genetic diseases that affect children globally, DMD causes more than 90% of individuals being confined to a wheelchair by age 15 (“Duchenne muscular dystrophy: The basics,” 2019). The burden of the disease is severe, with DMD being diagnosed in 1 among 3,500 to 5,000 males around the world (“Duchenne muscular dystrophy: The basics,” 2019). The statistics on the disease show that it has an adverse effect on the young male population.
The principal symptom of DMD is muscle weakness, which can begin in patients as early as at ages 2-3 (Birnkrant et al., 2018). The proximal muscles get affected first due to the fact that they are the closest to body’s core. The distal limb muscles get affected later because they are close to the body’s extremities (Birnkrant et al., 2018). Furthermore, it is notable that lower external muscles usually show DMD symptoms prior to the upper ones. In affected children, there are noticeable difficulties walking, running, and jumping, which decreases the quality of movement. Other symptoms of the disease include calves’ enlargement, waddle in the gait, as well as lumbar lordosis (Birnkrant et al., 2018).
The latter is a DMD sign implying an inward spine curve. Due to the difficulties associated with muscle strength, the affected children develop issues with the heart and respiratory muscles. The progressive weakness in the body, coupled with scoliosis, can lead to the development of impaired pulmonary function that causes acute respiratory failure.
When exploring the possible symptoms of DMD in their children, parents should understand that muscle deterioration may not be as painful as it is. This is because muscular dystrophy does not have a direct effect on the nerves, with touch and other senses being normal. This is also true for the functions of the bladder and the bowel. Furthermore, it is essential to note the development of learning disabilities among children with DMD.
While serious cognitive disabilities are rare, dystrophin abnormalities in the brain can have minor effects on both cognition and behavior (“Duchenne muscular dystrophy: The basics,” 2019). Learning problems can occur with focusing attention, memory and verbal learning, as well as emotional interactions. Children with learning disabilities who have DMD are usually evaluated by developmental or pediatric neuropsychologists who provide referrals to special education departments. Therefore, the manifestations of DMD can vary from developmental to physical impairments, which points to the need for managing the complications from the point of a multi-dimensional approach.
Genetic Issues
The underlying genetic defect that causes the condition’s development occurs in the dystrophin gene with an X-linked trait. This means that there is a mutation, an error, in one of the body’s genes that causes the DMD diagnosis. The dystrophin gene contains 79 exons, which are connected to create instructions intended for forming dystrophin protein, which is needed for muscle development (“Genetic testing: A Duchenne fingerprint,” 2019).
Within DMD, there may be three forms of genetic mutations. The first type of mutation is concerned with large deletions, with one and more exons missing from the dystrophin gene. The second type is concerned with large duplications, which means that one or more exons create copies of one another within the dystrophin gene. The third type refers to other changes in the gene, with small alterations taking place and ranging from deletions to changes in a single letter in a gene makeup.
As seen in the diagram below, large deletions of one or more exons in the gene (Figure 1). Similar to a puzzle, the missing pieces in the gene prevent the remaining exons from properly fitting together. This causes errors in the instructions for making dystrophin, with the body not being able to produce the necessary amount of dystrophin protein.
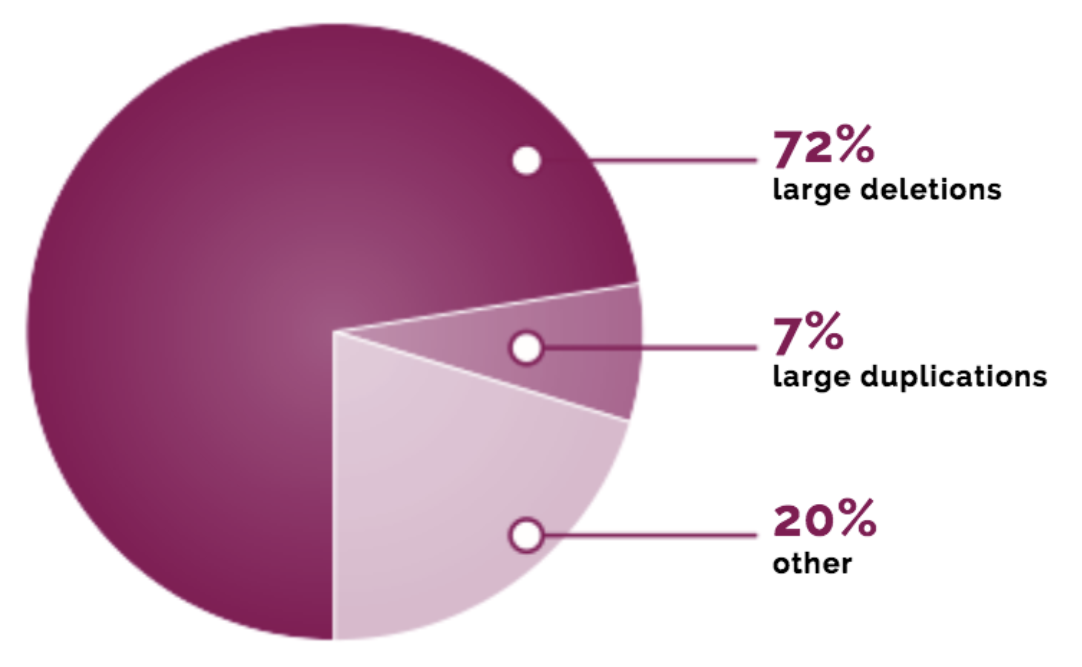
Understanding the importance of genetic mutations as related to DMD is essential because scholars have recorded more than 1,800 different changes in people’s diagnoses with either Duchenne or Becker types of muscular dystrophy. Knowing the kind of mutation that has occurred in a child is a fundamental step for considering the types of management (Bendixen & Houtrow, 2017). In order to determine the kind of genetic change, doctors prescribe a genetic test that provides further information on disease management. Moreover, children can be subjected to clinical trials that are currently being conducted to develop innovative treatments.
Different methods of genetic testing can be used for getting a full picture of a child’s mutation. For instance, complete gene sequencing can help to reveal small modifications in a gene. It is also important to note that since Duchenne is a genetic disorder, it can be inherited from one family member to another. The characteristic of DMD as an X-linked disease means that mutations are only found in the X chromosome (“Genetic testing: A Duchenne fingerprint,” 2019).
Therefore, in cases when women have Duchenne-causing mutations in their chromosomes, they are considered carriers of the disease. Carriers will most likely have no symptoms of DMD but will be capable of passing the gene along to a child; although, there is no certainty that the mutation will be transferred. The chance of having a boy from Duchenne from a carrier mother is 25%, with the same likelihood of 25% applied to having a carrier girl (“Genetic testing: A Duchenne fingerprint,” 2019).
This means that there is a 50% chance that a mother-carrier will have a baby with no mutation at all. In case when Duchenne is under suspicion, genetic testing is recommended, including carrier testing, in order to provide valuable information for making further decisions on treatment. Besides, the expertise of a genetic counselor can be highly helpful for explaining what genetic results on Duchenne can mean for families.
Diagnostic Process, Medical Management, and Prognosis
As mentioned in the previous sections, Duchenne muscular dystrophy is a rare inherited disease of the neuromuscular system that has no known cure at this time. DMD is usually suspected in mostly boys who display abnormal gait patterns and complications with physical activity, such as running or climbing stairs. Despite the fact that the current diagnostic process is straightforward, supported with more than 3 decades of research, there is still no solution to overcoming the healthcare challenge (Bendixen & Houtrow, 2017).
Furthermore, the American Academy of Pediatrics (2015) underlines the importance of addressing the concerns of parents about children’s development as soon as possible, especially in cases of more progressive and pronounced motor delays. Getting a formal DMD diagnosis is imperative for understanding a specific genetic mutation and determining an appropriate care path.
When a family pediatrician is concerned with the possibility of a DMD diagnosis in a child, a recommendation and referral for testing are made. An expert in neuromuscular management or a pediatric neurologist will further work to identify the causes of the symptoms and prescribe appropriate genetic and non-genetic testing.
Thus, the typical steps in identifying the disease include observing the signs and symptoms, conducting blood tests for determining enzyme levels (including CK test), genetic testing, and muscle biopsy when needed (Bendixen & Houtrow, 2017). A CK test will measure the blood’s concentration of creatine kinase, which would signify damage in muscle cells (Bendixen & Houtrow, 2017). The high levels of the enzyme in the blood will point to a muscle problem, but not confirm Duchenne as a final diagnosis as the testing is usually the first step.
Genetic testing is prescribed when an elevated level of CK is found, which means that Duchenne is suspected. This test will analyze the genetic makeup of an individual to determine a change in the dystrophin gene (Bendixen & Houtrow, 2017). When such a modification is identified, further decisions on management are made. A muscle biopsy may be prescribed when there is not enough certainty provided by a genetic test. While most patients do not require the biopsy, it is used for gathering more information.
Since there is no treatment for overcoming muscular dystrophy completely, some preventative and management efforts are taken to reduce the burden of disease. Treatment options predominantly range from medications to surgical procedures that help patients live with DMD and manage the condition’s symptoms. Doctors can prescribe heart medications and corticosteroids, as well as the Food and Drug Administration approved a drug called Eteplirsen (Exondys 51) (Bendixen & Houtrow, 2017). Despite being approved and safe for use, the drug has not shown significant evidence of effectiveness. While it may not cure DMD, it has the potential of improving muscle strength through acting on specific gene variants.
Corticosteroids are sometimes necessary for strengthening the muscle mass and delaying the development of certain types of dystrophy in children. The adverse effects of using corticosteroids over prolonged time periods include weight gain and weakened bone integrity, which increases the risk of fracture. Medications for the heart, such as angiotensin-converting enzyme (ACE) inhibitors and beta blockers, are usually prescribed when muscular dystrophy causes damage to the heart (Bourke et al., 2018). Overall, despite being prescribed to patients with DMD, medications cannot guarantee long-term maintenance of relative well-being.
Therapy and assistive devices are used as additional methods for managing life with DMD. Children diagnosed with the condition may do range-of-motion and stretching exercises to maintain high levels of mobility and flexibility of muscles as the disease causes limbs to be drawn forward and maintain in such a position (Magee, Zachazewski, Quillen, & Manske, 2015). Low-impact aerobic exercises that range from swimming to walking can help patients maintain the general health and mobility of children. Although, any activities should be verified with a doctor first since they can also be damaging. Braces and mobility aids are supplementary tools that maintain mobility independence and muscle stretchiness (Magee et at., 2015).
Breathing assistance is implemented when children’s respiratory muscles weaken. Severe cases of muscular dystrophy may call for the use of a ventilator that would force the air to travel to and from the lungs. It is also important to note that Duchenne may call for a surgery that would improve spiral curvature that limits breathing.
At this time, the prognosis for patients diagnosed with DMD does not include recovery. The adverse impact on children’s health causes a significant deterioration in life quality, leading to the need to monitor vitals continuously. For example, parents should always be aware of respiratory infection risks in more severe stages of muscular dystrophy in their children. Proper nutrition is also necessary to help prevent obesity, dehydration, and bowel movement issues. Seeking support from communities to help families cope with DMD contributes to the increased chances of living with the condition (Magee et at., 2015).
Therefore, psychological assistance is as valuable as physical management of the disease when it comes to complex genetic limitations such as Duchenne muscular dystrophy. Since DMD causes individuals with the condition to die the latest in their early twenties, extensive research is necessary to develop treatments that would be effective in overcoming the disease.
Concluding Remarks
Duchenne muscular dystrophy presents a significant challenge for the health care industry because of its severe impact on the well-being and development of children. Since DMD has no cure at the present time, it requires significant control on the part of children’s families and healthcare providers. While the combination of mild exercising and prescription medication can help patients deal with the disorder, the absence of effective interventions and treatment calls for further research and clinical trials.
References
American Academy of Pediatrics. (2015). Pediatric clinical practice guidelines & policies: A compendium of evidence-based research for pediatric practice. Elk Grove Village, IL: AAP.
Bendixen, R. M., & Houtrow, A. (2017). Parental reflections on the diagnostic process for Duchenne muscular dystrophy: A qualitative study. Journal of Pediatric Health care: Official Publication of National Association of Pediatric Nurse Associates & Practitioners, 31(3), 285-292.
Birnkrant, D. J., Bushby, K., Bann, C. M., Apkon, S. D., Blackwell, A., Brumbaugh, D., … DMD Care Considerations Working Group (2018). Diagnosis and management of Duchenne muscular dystrophy, part 1: Diagnosis, and neuromuscular, rehabilitation, endocrine, and gastrointestinal and nutritional management. The Lancet. Neurology, 17(3), 251-267.
Bourke, J. P., Watson, G., Muntoni, F., Spinty, S., Roper, H., Guglieri, M., … DMD Heart Protection study group (2018). Randomized placebo-controlled trial of combination ACE inhibitor and beta-blocker therapy to prevent cardiomyopathy in children with Duchenne muscular dystrophy? (DMD Heart Protection Study): A protocol study. BMJ Open, 8(12), e022572.
Duchenne muscular dystrophy: The basics. (2019). Web.
Genetic testing: A Duchenne fingerprint. (2019). Web.
Magee, D., Zachazewski, J., Quillen, W., & Manske, R. (2016). Pathology and intervention in musculoskeletal rehabilitation. Maryland Heights, MO: Elsevier.
Walter, M. C., & Reilich, P. (2017). Recent developments in Duchenne muscular dystrophy: facts and numbers. Journal of Cachexia, Sarcopenia and Muscle, 8(5), 681-685.