A 50-year-old married man with a family of three children presented chronic hepatitis C that developed to cirrhosis. Hepatitis C was linked to “intravenous drug used in 1960’s and trauma treatment involving several transfusion in 1975” (Schiff 2004, p. 183). Cirrhosis became complex due to “bleeding esophageal varice, but endoscopic banding intervention was successful in 2001” (Schiff 2004, p. 183).
The patient later had ascites and hepatic encephalopathy complications. A computed tomography conducted in “November 2001 showed a 6×6.5x7cm mass lesion in segment 7 of the liver (fig. 3), which later increased in size to 11.2×9.2×5.8cm on a repeat CT scan two months later while serum alpha-fetoprotein (AFP) was above 16,000ng/mL” (Schiff 2004, p. 183).
Introduction
Hepatocellular carcinoma (HCC) is the most “common main liver cancer and the fifth most widespread cancer worldwide with a poor prognosis and it is the third most prevalent cause of death from cancer” (Ghanaati et al 2012, p. 167; Hennedige, Tiffany and Venkatesh 2012, p. 530). Some studies have recorded that HCC is mainly common in Asia where there are rampant cases of hepatitis B and C infection.
The main risk factor for HCC is chronic hepatitis B virus infection (Carr 2012), which could be severe in densely populated regions (Ghanaati et al. 2012). Poor diet and metabolic factors may also influence prevalence of HCC (Abby and Zhu 2009).
The condition is common in men aged between 30 and 50 years of age (Yang and Roberts 2010). These authors also observe that HCC diagnosis mainly occur in advanced stages, which make it contribute to high incidence and cases of fatality. Moreover, this later diagnosis also reduces effective use of curative treatments.
Early diagnosis and surgical resection together with local ablation are effective in enhancing conditions of patients with HCC.
Imaging has critical role in determining the diagnosis of HCC. HCC has many imaging approaches, which include “ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET) and angiography” (Hennedige and Venkatesh 2012 p. 530; Ghanaati et al 2012).
These approaches are effective in diagnosis of patients with chronic liver problems and a possible HCC case. Imaging helps in “screening, diagnosis, medication planning, patient management, and observation after treatment” (Hennedige and Venkatesh 2012 p. 530).
In the case study, one can observed that HCC management is difficult because of the changing nature of the condition, factors that affect cirrhosis, and therapeutic challenges (Ghanaati et al. 2012). Surgical interventions and liver transplant have been effective in managing HCC. However, not all conditions of HCC may require surgical resection.
Thus, non-surgical interventions are common because of “multifocal diseases, proximity to the vital vascular or biliary structures and the insufficient functional hepatic reserve in cirrhosis” (Ghanaati et al. 2012 p.168).
In the absence of surgery, radiology options, such as transarterial chemoembolization (TACE), radiofrequency ablation, and microwave ablation (Firouznia, Ghanaati, Jalali and Shakiba 2011) are common in managing HCC (Schiff 2004).
Anatomy
The liver is the major and most multifaceted organ in the abdominal area of the body. It has several microscopic and functional lobules, which aid in various functions of the liver.
The liver originates from the ventral mesogastrium, but the upper section goes beyond the ventral area. Both falciform ligament and ligamentum teres connect the liver to the anterior section of the body while omentum joins the liver and the stomach as the coronary and triangular ligaments link the liver and the diaphragm.
The liver supplies blood to the body. It gets blood from the artery and vein. Hepatocytes are in charge for complex metabolic processes within the liver. In addition, they also account for processes related to lipid synthesis, carbohydrate and cholesterol control, secretion of lipoproteins, formation of urea, enzymes, serum albumin, and other proteins.
Moreover, the liver is critical in clearing drug chemical substances and unidentified bodies in it.
Kupffer cells aid in immune functions because they filter foreign particles in the liver. The liver also has biliary passages, which form bile and hepatic ducts. These ducts lead to the duodenum.
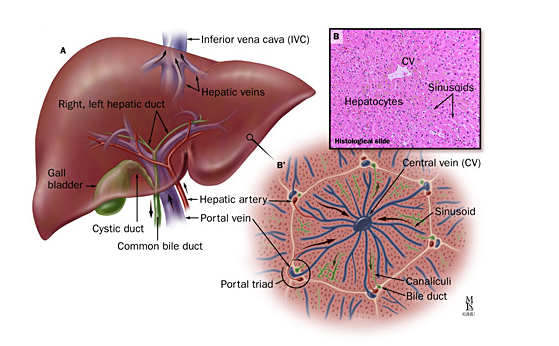
Figure 1: A: Normal Anatomy of the liver, B: Histological view (Gurakar et al 2013)
Pathology
HCC is also like other forms of cancer, which progress through various stages of tumour (Kojiro 2006). According to Sakamoto Masanori (2007), histopathological and molecular biological studies have revealed that HCC associated with “chronic liver disease evolves from precancerous lesions called adenomatous hyperplasia (also called dysplastic nodules) and early HCC to a progressed form” (Masanori 2007 p. 138).
During the early stages of the HCC condition, it looks like microinvasive carcinoma. Later the condition progresses to a progressed HCC. The early stage of HCC is critical for the development of all other subsequent stages (Sakamoto 2009).
In the case study, cirrhosis was in advanced stage, which resulted into several complications (Marrero 2003). However, it is difficult to understand progress processes of HCC during early stages, but recent studies have shown that HCC progresses from cirrhosis, dysplastic nodules to tumours (Marrero 2003).
Moreover, studies have failed to indicate any possible tumour suppressor genes during the early stages of HCC. Further studies in gene expression may clarify early stages of HCC. As cases of HCC rise, several combined tumours will be available and history of pathologic factors and patients’ history would be critical for understanding the condition.
Hepatitis B and C
Hepatitis B and C are fundamental conditions that contribute to HCC. There are several cases of HCC in China and Japan because of hepatitis B. In addition, other regions also experience cases of HCC from hepatitis B and C. While the prevalence of the condition is similar, many cases of HCC result from hepatitis C more than hepatitis B (Gomaa, Khan, Toledano, Waked, and Taylor-Robinson 2008).
Pathogenesis of HCC availability due to hepatitis B could result from high number of cell increment from chronic liver conditions or due to processes associated with hepatitis B viruses (Gomaa et al 2008).
Hepatitis B DNA genome may interfere with the regulatory roles of the liver and increase the condition. On the other hand, details on how hepatitis C contributes to the progression of HCC remain unclear, but other studies allude that such patients could have suffered hepatitis B prior to hepatitis C (Gomaa et al 2008).
Cirrhosis
The case study shows that regardless of cirrhosis etiology, it is always a risk factor for growth and progression of HCC. Cirrhosis has higher chances of increasing the development of HCC than hepatitis in any population (Gomaa et al 2008). As hepatocellular progresses, it may cause changes in tumour suppressor genes. Mutation may cause hepatocarcinogeneses. Majority of patients with HCC also have cirrhosis conditions (Carr 2012).
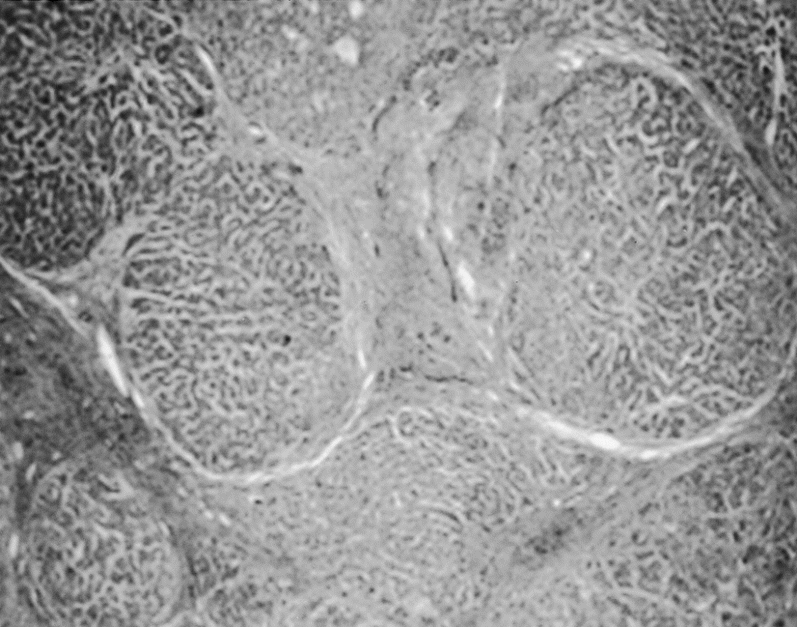
Figure 2: Liver biopsy showing presence of cirrhosis, (Schiff 2004)
Other Factors
There are also a number of etiological factors, which affect HCC. They include alcohol, use of anabolic steroid, aflatoxins, and hemochromatosis (Gomaa et al 2008). Dietary compositions with carcinogenic aflatoxins are usually widespread in some parts of sub-Saharan Africa and Southeast Asia (Gomaa et al 2008). Consequently, there is high prevalence of hepatitis B in these regions.
However, details on how hepatitis B and carcinogenic aflatoxins contribute to HCC remain poorly studied and understood (Gomaa et al 2008). In cases of hepatitis C infection, alcohol has played a major role in enhancing the condition of HCC. Still, it is not clear whether alcohol acts independently, or it acts with hepatitis C to increase the condition of HCC (Gomaa et al 2008).
Role of imaging modalities in the diagnosis
Imaging modalities include “ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET) and angiography” (Ghanaati et al. 2012). These modalities are useful in diagnosis of severe cases of liver conditions or suspected HCC (Ghariba, Thomasson and Li 2004).
Ultrasonography (US)
Ultrasonography is the first and commonly accepted form of HCC diagnosis because it is safe, fast, and cost-effective (Lee, Yoon and Kim 2012). However, ultrasonography does not provide optimal image quality in patients with cirrhosis. In this case, other modalities like CT and MRI are effective.
The new ultrasound elastrography relies on strain to identify mechanical elements of tissue stiffness during diagnosis. The method may not be possible to apply because of deep and protected location of the liver. In this case, acoustic radiation is necessary in order to improve impulse elastography with short and focused US beam for tissue displacement (Ghanaati et al. 2012).
Elastography has the ability to diagnose stiff superficial tissues, but “acoustic radiation force elastography can estimate the stiffness of the superficial and deep tissues due to application of shear wave” (Choi 2010). This method works well together with other conventional ultrasonography because it enhances conspicuity and differentiates between benign and malignant lesions (Hosseini 2011).
Computed tomography (CT)
CT images are useful in diagnosis of central liver lesions. This may take place after sonography and normal sonograhphy for patients who may have high-levels of α-fetoprotein (Sahani, Holalkere, Mueller and Zhu 2007).
In the case study, CT imaging showed that the patient had a 6×6.5x7cm mass lesion in segment 7 of the liver (fig. 3), which later increased in size to 11.2×9.2×5.8cm on a repeat CT scan two months later while serum alpha-fetoprotein (AFP) was above 16,000ng/mL (Schiff 2004; Sahani, Holalkere, Mueller and Zhu 2007).
CT scan serves different roles during diagnosis. It is applicable during “HCC staging and post-diagnosis after surgical resection, percutaneous ethanol injection, or radiofrequency ablation” (Ghanaati et al. 2012). CT scan improves contrast in HCC diagnosis. This happens after an injection of extracellular contrast-enhancing agent, which aid in depicting and identification of HCC.
CT perfusion performance involves the use of Multi Detector CT (MDCT) and injection of iodinated contrast materials in order to enhance temporary changes in tissue attenuation (Ghanaati et al. 2012).
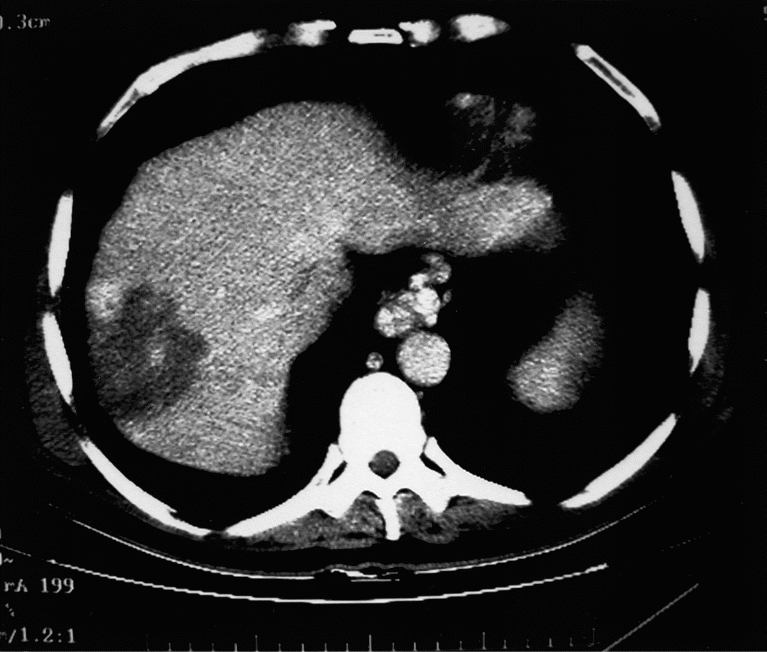
Figure 3: CT scan demonstrating mass in right hepatic lobe (Schiff 2004)
Magnetic resonance imaging (MRI)
MRI is suitable for patients who have not shown any diagnostic results in CT diagnosis or patients who may not show positive results with iodinated contrast agents. MRI is better than CT scan in cases of hyperintense malignant lesions. In addition, enhanced MRI yields better images than CT contrast-enhanced scans. Still, MRI shows minute lesions clearly than other modalities of HCC diagnosis.
MRI produces better result and efficacy relative to CT scan for effective decision-making on managing patients’ conditions. There is also Magnetic resonance elastography (MRE), which does not attack the body tissues, but reviews mechanical qualities of the body tissue. MRE aids in identification of malignant and benign liver tumours (Choi 2010).
MRI uses Diffusion-weighted image (DWI) technology as a non-invasive quantification to enhance the rate of detection of focal liver conditions (Ghanaati et al. 2012). The technique uses cellular necrosis to enhance membrane permeability to facilitate free diffusion of water molecules.
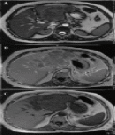
Figure 4: MRI HCC (Ghanaati et al 2012)
Positron emission tomography (PET)
PET is a new modality of HCC diagnosis. However, it is a new technique in terms of functions in HCC diagnosis, particularly in tracing certain hepatocyte. This technique will eventually improve the HCC diagnosis among patients with severe conditions of HCC and liver problems.
PET technique yields a three-dimensional image of the body (Bailey, Townsend, Valk and Maisey 2005). It uses gamma rays from tracer in order to construct the image. PET can also use CT scan to accomplish the imaging process.
Treatment and prognosis
Studies have indicated that HCC is not sensitive to radiotherapy or chemotherapy interventions.
However, several interventions exist for managing HCC, which include cisplatin gel injection, percutaneous ethanol, cryosurgery, radiofrequency ablation (surgical and percutaneous), hepatic artery chemoembolization, and liver transplant among others (El-Seragemail, Marrero, Rudolph and Reddy 2008). However, the case study focuses on the RFA and chemoembolization for managing advanced conditions.
Table 1: Barcelona Clinic Liver Cancer (BCLC) staging classification and treatment schedule
Adapted from (Sala, Forner, Varela and Bruix 2005)
Radiofrequency Ablation (RFA)
RFA is a relative new method of managing HCC. It relies on ‘heating’ to destroy tumour in the liver. It has a thin probe, which reaches the tumour with the aid of ultrasound (Bruix and Sherman 2005).
However, this technology applies to small tumour within the liver. Moreover, it has low-levels of side effects to the HCC patient. RFA is also simple to use because it can work in “an open or laparoscopic technique, physicians can apply RFA by the use of percutaneous approach (a direct injection through the skin)” (Gurakar et al 2013).
Physicians can also use RFA alongside other interventions like liver resection and cryotherapy (Ghanaati et al. 2012).
Hepatic Artery Chemoembolization
This is the most common intervention method for treating unresectable liver tumours (tumours beyond operation). Chemoembolization treatment offers many benefits than other intravenous pump infusion interventions (Bruix, Sala and Llovet 2004).
It takes the injected drugs to the tumour and controls blood flow without extending contact time for tumour and drug. It reduces oxygen supply to the tumour and improves high drug concentration on the tumour. Chemoembolization also has a long duration for drugs to stay in the liver, which may be more than a month after the treatment. It also enhances high chances of the patient survival after undergoing medication.
Chemoembolization has high-levels of vascularity, which enhances neuroendocrine tumour treatments. In fact, Chemoembolization is the most favourable intervention for unresectable hepatocellular carcinoma, and neuroendocrine tumours of the liver (Bruix et al 2004).

Figure 5: Liver tissue chemoembolization (Schiff 2004)
Summary and conclusion
HCC is a common primary liver cancer with poor prognosis and treatment, which have contributed to high numbers of mortality rate associated with it. The condition progresses through various stages as the patient in the case study indicates.
Studies have linked HCC with hepatitis B and C, liver cirrhosis and other factors like alcohol, use of anabolic steroid, aflatoxins, and hemochromatosis. However, details on how these factors relate to enhance HCC remain scanty.
Imaging techniques (specifically CT and MRI) have critical role in the diagnosis and evaluation of liver problems, tumours, and HCC interventions. Imaging helps patients whose conditions do not require surgery. The patient in the case study required chemoembolization because liver transplantation was not effective due to high chances of recurrence and the intermediate stage of the condition.
Reference List
Abby, Siegel and Andrew X. Zhu. “Metabolic Syndrome and hepatocellular carcinoma.” Cancer (ACS) 115, no. 24 (2009): 5651–5661. doi:10.1002/cncr.24687.
Bailey, Dale, David W. Townsend, Peter E. Valk and Michael N. Maisey. Positron Emission Tomography: Basic Sciences. Secaucus, NJ: Springer-Verlag, 2005.
Bruix, Jordi and Morris Sherman. “Management of hepatocellular carcinoma.” Hepatology 42, no. 5 (2005): 1208–1236. doi:10.1002/hep.20933.
Bruix, Jordi, Margarita Sala and Josep Llovet. “Chemoembolization for hepatocellular carcinoma.” Gastroenterology 127, no. 5 (2004): S179–S188. doi.org/10.1053/j.gastro.2004.09.032.
Carr, Brian. “Introduction: Hepatocellular Carcinoma.” Seminars in Oncology 39 no. 4, (2012): 367–368.
Choi, Bi. “Advances of imaging for hepatocellular carcinoma.” Oncology 78, no. 1 (2010): 46–52. doi: 10.1159/000315230.
El-Seragemail, Hashem, Jorge Marrero, Lenhard Rudolph and Rajender Reddy. “Diagnosis and Treatment of Hepatocellular Carcinoma.” Gastroenterology 134, no. 6 (2008): 1752-1763. doi:10.1053/j.gastro.2008.02.090.
Firouznia, Kavous, Hossein Ghanaati, Amir Hossein Jalali, and Madjid Shakiba. “Advances of interventional radiology in treatment of hepatobiliary diseases in Iran.” Hepat Mon. 11, no. 7 (2011): 507–10.
Ghanaati, Hossein, Seyed Moayed Alavian, Ali Jafarian, Nasser Ebrahimi Daryani, Mohsen Nassiri-Toosi, Amir Hossein Jalali, and Madjid Shakiba. “Imaging and Imaging-Guided Interventions in the Diagnosis and Management of Hepatocellular Carcinoma (HCC)-Review of Evidence.” Iran J Radiol 9, no. 4 (2012): 167–177. doi: 10.5812/iranjradiol.8242.
Ghariba, Ahmed, David Thomasson and King Li. “Molecular imaging of hepatocellular carcinoma.” Gastroenterology 127, no. 5 (2004): S153–S158. doi.org/10.1053/j.gastro.2004.09.029.
Gomaa, Asmaa Ibrahim, Shahid A Khan, Mireille B Toledano, Imam Waked, and Simon D Taylor-Robinson. “Hepatocellular carcinoma: Epidemiology, risk factors and pathogenesis.” World J Gastroenterol., 14, no. 27 (2008): 4300–4308. doi: 10.3748/wjg.14.4300.
Hennedige, Tiffany and Sudhakar Kundapur Venkatesh. “Review: Imaging of hepatocellular carcinoma: diagnosis, staging and treatment monitoring.” The Journal of the International Cancer Imaging Society 12, no. 3 (2012): 530-547. DOI: 10.1102/1470-7330.2012.0044.
Hosseini, Saiedi. “Risk factors and incidence of hepatocellular carcinoma in southeast iran.” Hepat Mon. 11, no. 8 (2011): 666–7. doi: 10.5812/kowsar.1735143X.710.
Kojiro, Masamichi. Pathology of Hepatocellular Carcinoma. Oxford: Wiley-Blackwell, 2006.
Lee, Jeong, Jeong-Hee Yoon, and Kyung Kim. “Diagnosis of Hepatocellular Carcinoma: Newer Radiological Tools.” Seminars in Oncology 39, no. 4 (2012): 399–409.
Marrero, Jorge. “Hepatocellular Carcinoma:” Current Opinion in Gastroenterology, 19, no. 3 (2003): 243-249.
Sahani, Dushyant, Nagaraj-Setty Holalkere, Peter Mueller, and Andrew Zhu. “Advanced Hepatocellular Carcinoma: CT Perfusion of Liver and Tumor Tissue—Initial Experience.” Radiology 243, (2007): 736-743. doi: 10.1148/radiol.2433052020.
Sakamoto, Masanori. “Pathology of early hepatocellular carcinoma.” Hepatol Research 37, no. 2 (2007): S135-8.
Sakamoto, Masanori. “Early HCC: diagnosis and molecular markers.” Journal of Gastroenterology 44, no. 19 (2009): 108-11. doi: 10.1007/s00535-008-2245-y.
Sala, Margarita, Alejandro Forner, Maria Varela and Jordi Bruix. “Prognostic Prediction in Patients with Hepatocellular Carcinoma.” Seminars in Liver Disease 25, no. 2 (2005): 171-180.
Schiff, Eugene R. “Case Study in the Management of Patients with Hepatocellular Carcinoma.” In Management of Patients with Viral Hepatitis, Paris, 2004, edited by Fabien Zoulim, 183-188. Paris: Beaujon Hospital, 2004.
Yang, Ju and Lewis Roberts. “Epidemiology and management of hepatocellular carcinoma.” Infect Dis Clin North Am. 24, no. 4 (2010): 899-919. doi: 10.1016/j.idc.2010.07.004.