Introduction
The change from freezing winters to scorching summers that I experienced growing up in an Eastern European nation with distinct seasons reminded me of the divergent behavior of particles in various temperatures. This comparison, similar to Hess’s law in physics, illustrates how liquids and gases are more dynamic, akin to the life of spring and summer. In contrast, solids are more akin to the calm of winter, with particles held together.
This also made me want to investigate why the damper spring on my bicycle felt stiff and rigid during the winter and flexible and comfortable during the summer. I figured that this IA would be the best way to investigate how spring constants are affected by different temperatures of the spring by experimenting with which I will be placing a spring in different temperatures of water.
This experiment provides a compelling example of how physics can be applied in real-world situations. Springs are present everywhere and come in a variety of shapes and sizes. They are used as dampers, mattress components, and even as battery connectors in remote controls.
The robustness of these springs must be evaluated in various environmental conditions, including extreme temperatures. Understanding how they respond to different thermal stressors not only improves product dependability but also guarantees functioning and safety. This study highlights the pervasive influence of scientific knowledge on our contemporary environment by shedding light on how fundamental principles of physics affect the durability and utility of everyday objects.
Background Theory
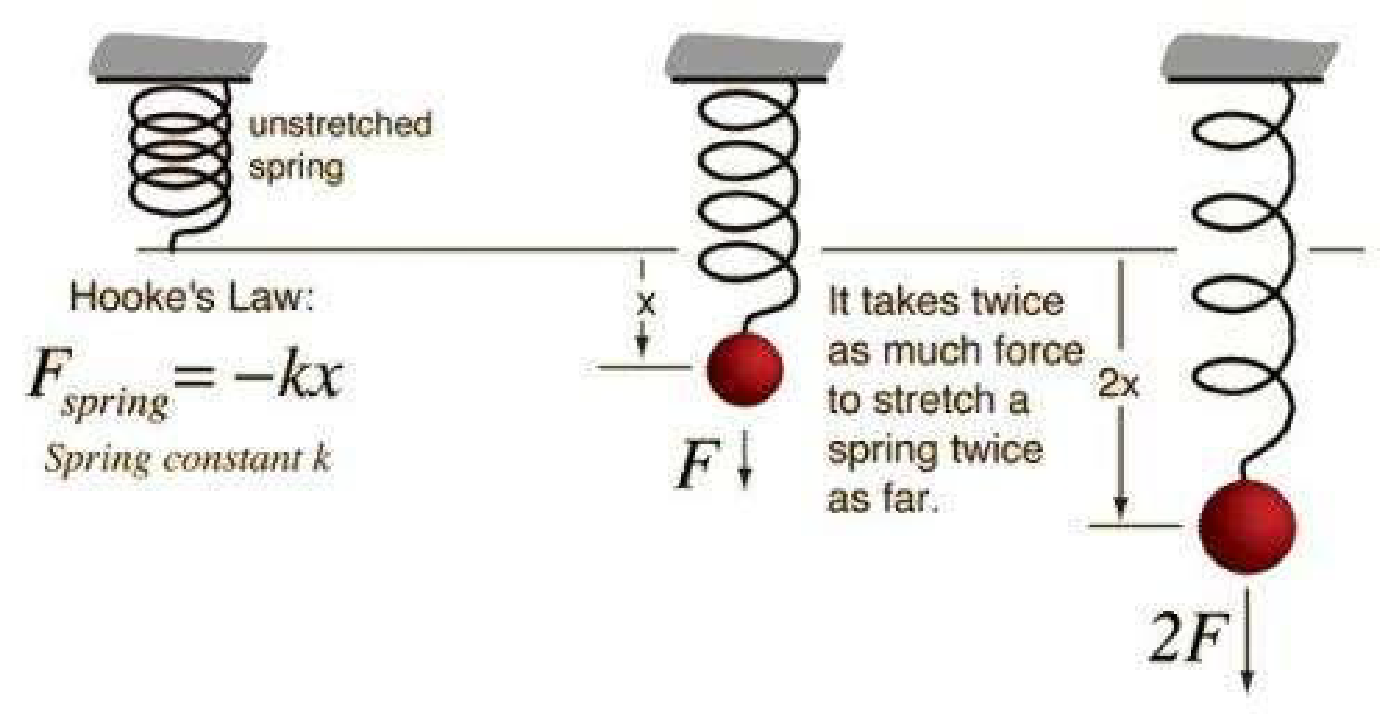
Regardless of how they are used in our daily lives, springs all follow the fundamental physical rule F = kx. The connection between force (F) and displacement (X) in the setting of springs is accurately defined by this equation (Bolmin et al., 2021). In essence, it means that a spring will produce an opposing force that is directly proportionate to the amount by which it has been compressed or extended. For instance, if the spring is stretched twice as far, it will generate twice as much force, and this proportionality applies to different levels of extension. As a measure of a spring’s elasticity, the spring constant, denoted by “k,” is used.
Depending on the material or thickness of the spring in issue, this constant may change. When “X” remains constant, according to Hooke’s law, both “F” and “k” increase in proportion. Practically speaking, this means that springs with greater stiffness have larger spring constants. Unfortunately, the spring constant cannot be directly measured, which would have made things easier. So, using Hooke’s rule to measure both “F” and “X,” I can finally determine “k” by applying it.
Hypothesis
The experiment’s water temperature will rise, and there will be a detectable drop in the spring’s spring constant (k). The well-known correlation between temperature and the kinetic energy of particles in a material predicts this event. The average kinetic energy of particles within the spring’s material increases as the temperature rises, in accordance with the kinetic theory of matter (Oyelade, 2020). Mathematically, this rise in kinetic energy is related to a fall in the spring constant, which reflects a rise in the spring stiffness. Although the change in spring constant may not be significant, it is anticipated to be observed within the parameters of this experiment.
Variables
The dependent variable in this experiment is the spring constant (K), which serves as the essential parameter we will measure to analyze the spring’s behavior. The temperature of both the water and the spring, on the other hand, is an independent variable. We intend to explore the effect of thermal energy on the spring constant by manipulating and adjusting the temperature levels. We anticipate witnessing variations in the spring’s elasticity as we systematically adjust the temperature, which will be quantified by the observed spring constant(Bolmin et al., 2021).
This experimental approach enables us to establish a cause-and-effect relationship between temperature and spring behavior, allowing us to gain a deeper understanding of material characteristics and their response to thermal variations. Any scientific experiment must include control variables, commonly referred to as constant variables. For the experiment’s ultimate results to be reliable and accurate, they must stay constant and unchanging throughout. Researchers can isolate the effects of the independent variable by holding these variables constant. This prevents outside influences from introducing anomalies or confounding factors that could skew the results of their investigation, improving the overall validity and robustness of their findings.
The reliability and correctness of the experiment depend on the standardization of several crucial variables. The selection of the spring is crucial since the primary variable under research is its stiffness, which is defined by the spring constant. We must use the same spring throughout the experiment to ensure consistency and reliability, as different springs may have varying spring constants, which could skew our results.
The technique by which we assess the spring is equally important as its constant usage (Wulandari et al., 2021). To ensure that our measurements are taken from the same reference point each time, we will consistently measure from the first coil to the bottom of the spring, thereby maintaining accuracy and consistency. This rigorous measurement technique helps eliminate potential data errors.
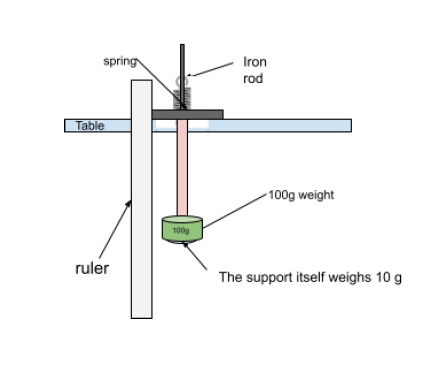
Additionally, the force applied to the spring before and during heating is crucial to the experiment’s precision. I will continually apply a weight of 110 grams to the spring during each trial to ensure homogeneity in the applied force, preventing variations in force. Finally, the heating technique needs to be closely monitored. We will employ a water bath and gradually raise the temperature in 10-degree Celsius increments, ranging from 25 to 45 degrees Celsius, to minimize unintentional overheating (Oyelade, 2020). This strategy ensures that we maintain the desired temperature range without risking harm or unintentional changes to the spring’s characteristics. We can conduct a thorough and trustworthy experiment to investigate the relationship between temperature and spring constant by rigorously standardizing these components.
Apparatus
- One dark-colored spring
- An infra-red thermometer
- A thermometer for cross-checking
- A water bath
- A 100g weight with a 10g hook
- A clamp
- A 30 cm ruler
- An iron stand
- Table
- Ring clamp.
Method
- Set the stand up.
- Heat the spring to the first temperature by putting it in the water bath and monitor the temperature with both thermometers for precision.
- Take the spring out and measure it by hanging it on the clamp, and record the values.
- Hang the mass to the spring and measure and record the new values.
- Take the mass off the spring.
- Heat the spring to the other numbers, then repeat steps 4-6 for each data point.
- Wait for the spring to cool down to its original temperature and then repeat steps 4-7, three times for each value.
One factor that is particularly beneficial about the method I will be using is that it ensures even heating. This means that all the parts of the spring will have the same temperature, which reduces the random error.
Safety and Environmental Issues
Considering we are not working with natural ecosystems or live creatures, safety and ethical issues in this experiment are very simple. As a result, no ethical concerns are raised by the potential harm to animals or the environment. The handling of hot water is what receives the most attention from a safety standpoint.
There is a potential danger of burns or scalds when the water in the experiment is warmer than the temperature considered pleasant, which is typically an average of 40 degrees Celsius, according to conventional recommendations. To minimize the risk of mishaps, certain safety precautions must be taken before removing the spring from the hot water bath. To prevent splashes or direct contact with hot water, use heat-resistant gloves or other protective equipment. Although there are no inherent ethical problems with the experiment, safety should always be the primary concern when dealing with high temperatures to prevent accidents or injury.
Data Collection and Processing
First, I will insert the table of raw data that I have collected, which will show the relationship between the displacement and the temperature of the spring after two minutes in the water bath (value of X in Hooke’s law). Then, I will use the equation to determine the spring constant (k value) for each of the x values and create a graph showing the relationship between displacement and the spring constant.
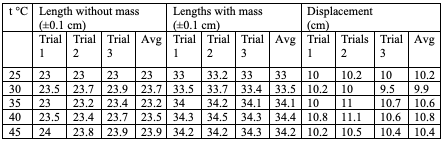
In determining the force F in Hooke’s law, F=kx. The sign is irrelevant when calculating this, as only the magnitudes are essential in this calculation. In our case, the force acting on our spring is the gravitational force exerted by the hanger with its mass. The weight of the mass was 100 grams, and the hanger weighed 10 grams; therefore,
m=100g+10g=0.100 kg + 0.010 kg= 0.110kg. Then the gravitational potential equation to find F can be used since we have the mass and the gravitational acceleration:
F=mg→ (0.110 kg) X (9.81 m/????) 1.08 N
I have not included any doubt in this calculation because I have taken the mass values as they were written on the mass and the hanger themselves.
Table 2. Spring constant for different temperatures
The uncertainty in the spring constant varies because F has no uncertainty, and it implies that the only vagueness valid for the value of k would be the same indecision that has been used for the value of x. Hence, the percentage ambiguity for the value of x is: which is quite a high hesitation. We can then calculate 2 cm × 10.2 cm = 19. 6% this percentage and multiply it by the mass of the mass plus the hanger, and that would give us a value of 19.6%X0.110=2.15 N/cm.
Errors, Limitations, and Improvements
In this experiment, the lack of previous theoretical work limits the ability to compare outcomes with existing data, thereby lowering the probability of systematic errors (Monteiro, 2022). Future studies’ accuracy and dependability can be improved in several ways, though.
Heating Device and Procedure
The use of a water bath presented difficulties in achieving consistent heating because of the sluggish temperature swings and the 45-degree Celsius temperature restriction. A Bunsen burner or another more effective heating source might be used to solve this problem. Rapid and accurate temperature management would enable a wider temperature range and more rapid transitions between values, leading to more exact measurements of the spring constant.
Measurement Time Relative to Temperature
Because the time it takes to clamp the spring back to the iron support might vary, random mistakes could be introduced during the spring’s cooling process after being withdrawn from the water bath. This might be mitigated by accelerating temperature changes using the earlier-mentioned heating technique optimization, which would shorten the time the spring has to cool outside the water. This source of mistake might be reduced even more by standardizing the clamping procedure.
Time of Heating
In the current experiment, the two minutes allotted for immersion in the water bath may not have been enough time for the spring to attain the appropriate temperature. A more extended heating period would guarantee that the spring and the water approach thermal equilibrium, decreasing temperature variations and improving the precision of measurements of the spring constant.
Limitations of the Experiment
This experiment has two drawbacks that prevent a thorough knowledge of spring constants. First, observations could only be made within a limited temperature range of 25°C to 45°C due to the heating method and time restrictions. A larger range could have shown more substantial changes in the spring constant. Second, because only one spring was used in the experiment, it was unable to determine how different initial spring constants would have affected the results. These restrictions highlight the importance of considering sample variety and equipment limitations when conducting experiments to gain a more comprehensive understanding of physical parameters, such as spring constants.
Conclusion and Analysis
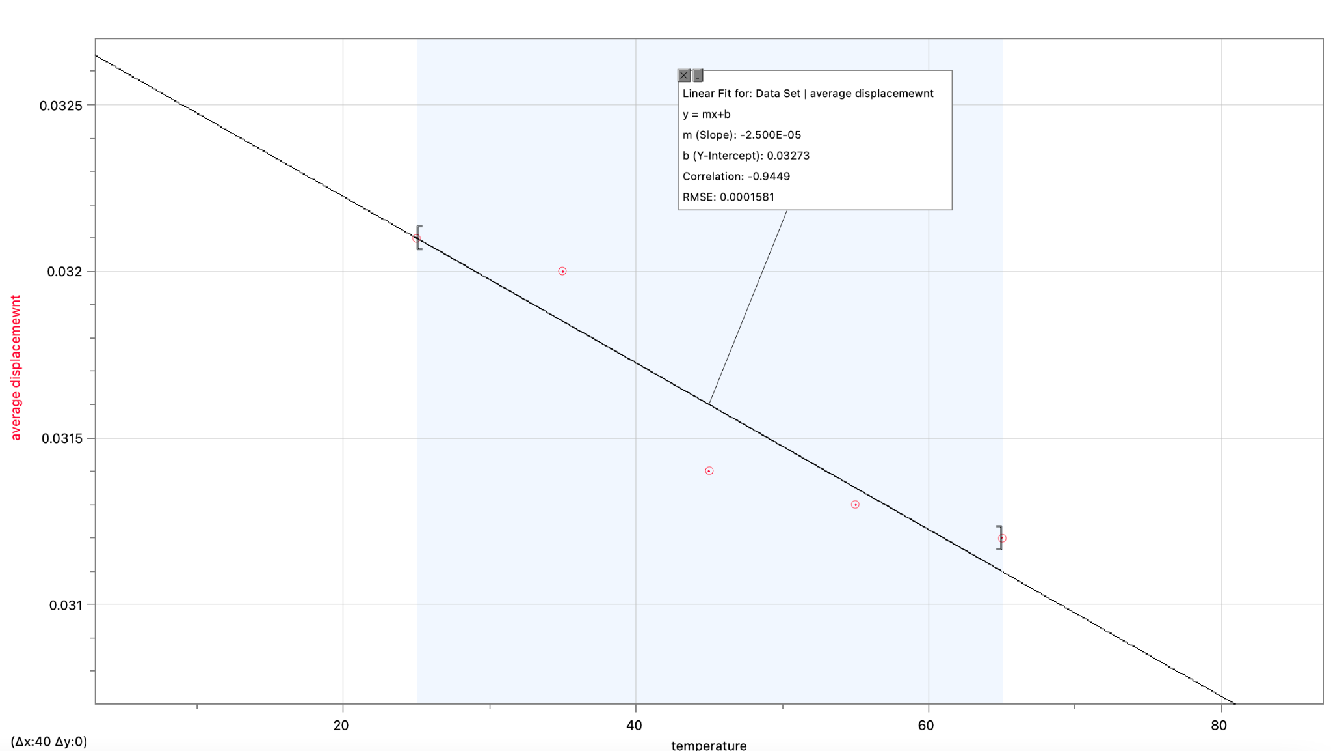
The purpose of this experiment was to examine how temperature affects a dark-colored spring’s spring constant (k). According to the theory, when the spring’s temperature rose, the spring constant would also drop since the particles inside the spring material would have more kinetic energy. The spring was submerged in water baths of varying temperatures during the experiment, and the displacement of the spring caused by a hanging mass was measured. It was clear from the data analysis and charting of the findings that the spring constant indeed showed a declining tendency as the temperature rose. The inverse association between temperature and spring stiffness was demonstrated by a negative proportionality constant (m = -2.5 N/°C), despite the minimal change in spring constant, which fell within the range of experimental error.
A high R-squared value (0.893), indicating a significant correlation between temperature and the spring constant, confirmed the validity of the data. This supported the theory and suggested that the experiment’s findings were precise and reliable. However, it is essential to acknowledge certain limitations. The heating technique and the water bath’s maximum temperature of 70°C, which prohibited observations at extremely high temperatures, limited the experiment’s temperature range.
Additionally, adopting a single spring might not take into consideration the differences in initial spring constants that several springs could have. Future iterations should consider enhancing the heating technique for more precise temperature control and extending the temperature range to increase the experiment’s validity. The link between temperature and spring constants in real-world circumstances may also be better understood by conducting tests with a variety of springs of different initial stiffnesses.
References
Bolmin, O., Socha, J. J., Alleyne, M., Dunn, A. C., Fezzaa, K., & Wissa, A. A. (2021). Nonlinear elasticity and damping govern ultrafast dynamics in click beetles. Proceedings of the National Academy of Sciences, 118(5), e2014569118.
Monteiro, I. (2022). On Hooke’s rule of nature. History of science and technology, 12(2), 249-261.
Oyelade, A. O. (2020). Experiment study on nonlinear oscillator containing magnetic spring with negative stiffness. International Journal of Non-Linear Mechanics, 120, 103396.
Wulandari, S., Iswanto, B. H., & Sugihartono, I. (2021). Determination of springs constant by Hooke’s law and simple harmonic motion experiment. In Journal of Physics: Conference Series, 2019(1), 012053. IOP Publishing.