Introduction
It appears that many people still believe today that cancer is caused by genetic mutations. Recent studies have discovered that while such mutations may, indeed, lead to cancer, they are not the only possible causes of it. In fact, it was found out that epigenetic factors may also lead to cancer. Understanding this enables a better comprehension of the aetiology of cancer, which can potentially help in cancer detection and prevention. Therefore, this paper explains in more detail what epigenetics and epigenome are, how changes in them might lead to cancer, and why knowing this may assist in averting this disease in the future.
Epigenetics and Epigenome
Epigenetics is a branch of biology which is concerned with studying the changes in the expression of genes that occur not due to the changes in the DNA itself, but because of the conditions in which an organism exists and develops. In other words, epigenetics is an area of study that is aimed at researching the phenotype of an organism, and only that phenotype which does not result from genetic changes of an organism.
A typical example is changes in chromosomes, which may have a considerable impact on the activity of genes and their expression. Other examples may include prions – infectious agents that are often simply a protein which is capable of self-replication inside a living organism, during which it causes damage to the host. Sometimes prion diseases result from such causes as an infectious agent affecting a susceptible area of a DNA and inducing self-replication, rather than the transcription of the DNA, which makes the disease epigenetic.
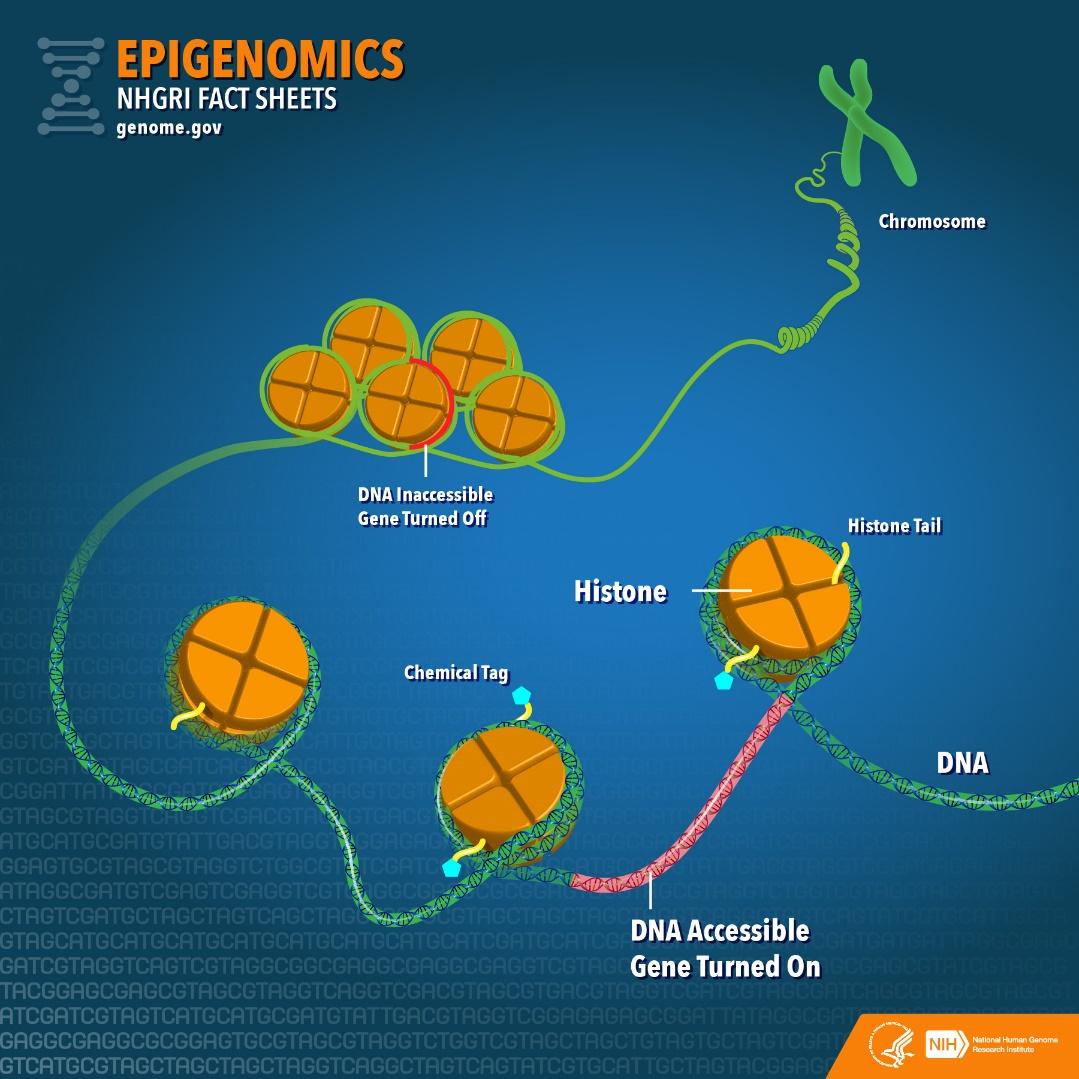
An epigenome is a set of chemical compounds which are capable of attaching to DNA in order to control it and modify its functions (for instance, to block the creation of some proteins and unblock the synthesis of other proteins), but which do not change the DNA (the gene sequence) itself. Epigenomes play a pivotal role in an organism, specifying what compounds need to be produced for which purposes. For instance, blocking and unblocking the creation of certain proteins is needed for cell specialisation.
More specifically, each cell in the organism possesses the same genome. However, when some proteins of the genome are blocked, and others unblocked, then, for instance, muscle cells are produced, whereas when yet other parts of the genome are activated, neurons are created. This makes the changes in epigenomes paramount for the proper development and functioning of any organism. It should also be stressed that epigenomes are usually not inherited, although some parts may be passed from parents to children.
Epigenomes can change both due to the natural needs of the organism (for instance, because of the necessity to produce specialised cells in response to a certain situation), and as a result of various environmental factors or some types of lifestyle (for instance, due to smoking, viral infection, or a certain diet). Some of the changes in the epigenome may be damaging, although the organism is usually able to withstand most adverse changes.
Cancer and Epigenetics
On the whole, cancer can be caused by genetic mutations, changes in the epigenome, as well as by a combination of both. Some cancerogenic changes in the epigenome may involve blocking or unblocking some genes which are responsible for the growth of cells, or for the immune response. Such changes have the potential of causing an uncontrolled growth of pathologic cells, or preventing the immune system from eradicating tumours.
Whereas genetic mutations may cause cancer in a rather straightforward manner (that is, by altering oncogenes and/or suppressors of tumours), the epigenetic changes that are observed in cancerous cells are considerably more complicated.
More specifically, they are determined by the chromatin structure, which includes such ways of affecting the activation of DNA as the DNA methylation, histone modifications, or alterations of non-coding regulatory ribonucleic acids, especially miRNAs (an abbreviation for a microRNA) (see Figure 1 below<). Therefore, when a tumour is initiated and begins developing, a variety of changes take place in the epigenome, most commonly, such changes as hypomethylation or hypermethylation, alterations in the occupancy of nucleosomes, and frequent rises in promoter methylation of CpG islands. Other changes are also possible, however.
In this respect, it should be noted that methylation is an attachment of the methyl group to the 5-methylcytosine nucleotide of the DNA, and it is usually associated with the function of gene silencing. Therefore, the term “hypomethylation” often refers to a loss of DNA methylation across the whole genome, although in certain cases it may also denote the loss of methylation in some local areas of the DNA. Considerable hypomethylation is an epigenetic change which is often found in cancerous cells, for instance, in melanomas, gastric or colorectal cancers, and others; with the progression of cancer, hypomethylation often increases.
Apart from hypomethylation, which “unsilences” genes that should be inactive, an opposite phenomenon, hypermethylation, is also often found in cancerous cells. It is critical to observe that hypermethylation is excessive methylation occurring in sites which do not require methylation for appropriate functioning. The excessive methylation causes gene silencing, and usually takes place in promoter CpG islands; it is associated with loss of gene function, and can most frequently be found in genes that are responsible for the suppression of tumours.
Some other epigenetic changes that can often be found in cancerous cells are serious modifications of histones, that is, of the proteins which serve as spools for DNAs to wind around, and which order DNAs in structural units(see Figure 1 below). Histones can often be subjected to modifications after changes are transferred to amino acids as a natural process. However, aberrant histone modifications were also found in cancerous cells, for instance, in patients with prostate cancer, nonsmall lung cancer, bladder cancer, and renal cell carcinoma.
Finally, the third main group of epigenetic changes that was found in cancerous tissues pertains to modifications in miRNA. It should be observed that miRNAs serve as controllers for target genes expression by having an impact on the stability, as well as on the transitional rate, of messenger RNA. Therefore, it turns out that miRNAs are also affected in cancerous cells; in fact, epigenetically silenced miRNAs were found in tissues associated with cancerous metastases. In addition, studies revealed that miRNAs in cells affected by breast cancer contained silenced tumour suppression sequences, whereas oncogenic miRNAs had excessive expression. Thus, such epigenetic changes as certain alterations in miRNAs reflect the progression of cancerous cells.
Thus, on the whole, it is possible to state that a serious enough failure of epigenomes (for instance, improper conservation of epigenetic marks) may become the cause of inappropriate blocking or activation of certain signalling pathways, which, in turn, can start the process of tumorigenesis. Generally speaking, cancerogenic epigenomes are often characterised as possessing considerable alterations in a variety of epigenetic regulatory layers. It is worth observing that epigenetic silencing of certain genes may result not only in the loss of function but also in an increased risk of malicious genetic mutations or changes in signalling pathways, which can also become a cancerogenic factor.
From the information provided above, it is also clear that epigenetics may allow for a better understanding of the aetiology of various types of cancer. Malfunctions which affect the correct manner of DNA transcriptions, such as improper methylation or histone modifications, block or otherwise prevent the activation of genomic sequences which are responsible for suppressing cancerous growth, whereas the genes which stimulate the proliferation of tumours are “unsilenced.” In addition, it is paramount to stress that epigenetic changes can also be responsible for introducing genetic mutations into the DNA, which may also be of a cancerogenic nature. Therefore, generally speaking, epigenetic changes often take advantage of genetic mutations or even induce them. So, while there are cancers which only result from harmful epigenetic alterations, many of them may be caused by both types of problems.
Epigenetics and Lung Cancer
On the whole, the epigenetic changes that were described in the previous section also pertain to various types of lung cancer. It is stated that lung cancer, similarly to other cancers, is induced by local methylation problems, which causes cancer-associated genes to become effective.
Numerous genes are cancer-associated promoters that are hypermethylated in cancerous cells. It is interesting that because of this DNA methylation can be utilised as a biomarker, allowing for early detection of lung cancer by analysing bodily fluids such as sputum, bronchoscopic materials, or even blood. Such tests should be more effective than the commonly used cytological examinations, which only allow for detecting lung cancer in only nearly half of all cases. Nevertheless, it can be pointed out that due to the lack of validation, it is still impossible to identify the exact potential of tests using methylation as biomarkers.
A study by Kwon et al. also explores the idea of using DNA methylation as biomarkers for detecting cancers, and lung cancer in particular. However, it is suggested that during studies, an entire human genome should be profiled, not only some of its areas; this allows for identifying more genes regulated by the mechanism of DNA methylation in the process of cancer development. Kwon et al. conclude that numerous genes had been previously demonstrated to be subjected to hypermethylation in patients with nonsmall cell lung cancer.
These scholars also identified three genes subjected to hypermethylation and one gene undergoing hypomethylation in human lung squamous cell carcinoma. In this regard, it should be stressed that identifying genes affected by abnormal methylation may potentially serve for both diagnostic and prognostic functions for cancers, including lung cancer.
Apart from DNA methylation in cancerous cells, miRNA also undergoes substantial changes. It is noteworthy that certain miRNAs were found to be associated with the invasiveness of tumours and the metastatic potential of a number of types of cancers, including lung cancers. Also, because miRNAs are estimated to be responsible for regulation of up to 30% of human genes, and due to their small size (which allows for their safer storage and lowered degradation), they are also expected to become useful as biomarkers for early detection of various types of cancer, in particular, lung cancer.
Finally, histone modifications in the context of lung cancer should also be discussed. Generally speaking, chromatin structure (i.e., DNA + histone) is paramount when it comes to determining if a particular gene will or will not be expressed. Condensed chromatin results in gene suppression, while genes that can be transcribed are located in the regions of open chromatin. Therefore, changes in histone may be associated with creation of pathogenic tissues, and studies indeed reported aberrant patterns of modification of histone in lung cancer. It is noteworthy that such modifications were different in patients at different stages of lung cancer, and that certain histone alterations were associated with better survival probabilities than other ones. This also allows for concluding that histone modifications may serve as a useful biomarker for identifying patients with cancer.
Conclusion
On the whole, it should be stressed that recent studies have discovered that epigenetic factors, i.e. mechanisms that are responsible for activating/deactivating certain parts of DNA to be transcribed may lead to cancer if the epigenome is aberrantly modified. This implies that:
- such aberrations might serve as biomarkers for early detection of cancers; and
- that they may potentially be repaired to prevent cancer.
However, both implications, especially the second one, require further research before they can be reliably applied in practice.
References
- Dawson, M. A. & Kouzarides, T. Cell 150, 12-27 (2012).
- National Human Genome Research Institute. NIH. Web.
- You, J. S. & Jones, P. A. Cancer Cell 22, 9-20 (2012).
- Sandoval, J. & Esteller, M. Curr. Opin. Genet. Dev. 22, 50-55 (2012).
- Smith, A. K. et al. Brain Behav Immun. 38, 227-236 (2014).
- Kang, K. A. et al. Tumour Biol. 33, 403-412 (2015).
- Lv, T. et al. PLoS One Web.
- Liloglou, T. et al. Cancer Lett. 342, 200-212 (2014).
- Langevin, S. M., Kratzke, R. A. & Kelsey, K. T. Transl Res. 165, 74-90 (2016).
- Kwon, Y.-J. et al. J Thorac Oncol. 7, 20-33 (2012).