Genzyme’s focus on orphan drugs affecting the degree of competition
The Genzyme company has used a strategy somewhat similar to the “blue ocean strategy”; although it did not create a unique market, it was able to provide rare products to a target market that did not have any significant rivals during the time. The company did have fewer clients, but the number of competitors reduced as well; insurance companies were also less likely to resist reimbursement (Schilling 2012). Due to high switching costs for customers (if any alternative was present at all), the bargaining power of customers was low. The drugs manufactured by Genzyme are nonstandard, rare, and difficult to obtain. As larger pharmacological companies are not interested in this niche market, Genzyme’s competitive advantage continues to increase.
Genzyme’s focus on orphan drugs affecting the needed types of resources and capabilities
As orphan drugs require specific resources (e.g., human placentas), biotech companies either need to cooperate with larger pharma firms or seek resources independently (as Genzyme did). The bargaining power of suppliers can increase in this case, as recourses for orphan drugs are rare, and the number of available suppliers is low. Genzyme’s decision was to handle resources independently, without the support of any large firms, which eventually contributed to its success in the market, as it was not as dependent on suppliers as smaller start-ups were (Köhler & Rammer 2012). At the same time, a company that does not cooperate with other firms can have fewer capabilities in conducting tests and invests less in research and development in general, which can undermine their success in the market.
Genzyme’s focus on orphan drugs as a long-term strategic intent
The long-term strategic intent of Genzyme is evident: it aims to focus on a small niche market that provides customers with high loyalty and is protected by government regulations and a small number of rivals. Using technology and innovation, it plans to profit from a small but steady number of customers and continue manufacturing medicine for rare diseases, thus expanding its target market. One of the most important aspects of targeting a niche market is specialization: “specialization is primarily related to the product knowledge and the ability to create unique and differentiated products” (Toften & Hammervoll 2013, p. 277). Genzyme’s specialization in products is clear; it continues to support research on potential customers and disease development, thus improving their knowledge about the target market and aligning it with their strategy.
Genzyme’s diversification into other areas of medicine
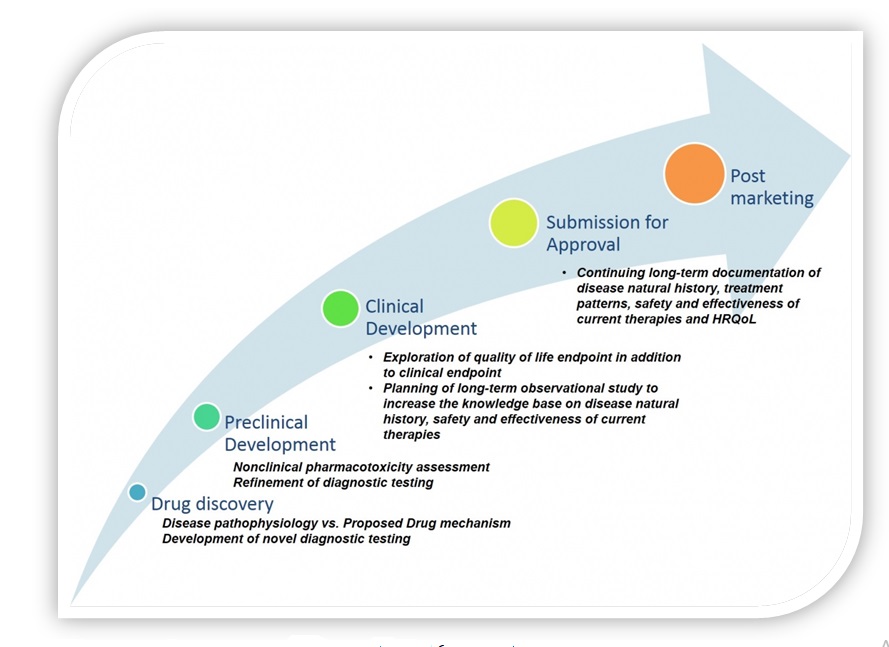
To remain independent, Genzyme had to generate revenues for future investments from other sources (not partnerships) and entered several side ventures (a chemical supplies business, a genetic counseling business, a diagnostic testing business) (Schilling 2012). Due to the complex process of orphan drug approval by the FDA (see Figure 1), diversification can be advantageous as it provides additional financial support for drug research, development, testing, and marketing. At the same time, any significant losses inside ventures will adversely influence Genzyme, leading to lower investments in technology and innovation, and increasing the competitive advantage of rivals.
Recommendations for Genzyme’s future
Genzyme can continue focusing on the orphan drug market but might expand its target market by addressing other rare medical conditions, e.g., autoimmune or endocrinological diseases. These markets are bigger, but the presence of medicine manufacturers for rare diseases in these areas is also small compared to “blockbuster” pharmacology. New target markets will support Genzyme’s independence from more prominent players. To penetrate these markets effectively, Genzyme will need to conduct extensive market research and segmentation, evaluate the competitive advantage of present rivals, and assess the bargaining power of suppliers and buyers.
Reference List
Köhler, C & Rammer, C 2012, ‘Buyer power and suppliers’ incentives to innovate’, ZEW Discussion Papers, vol. 12, no. 58, pp. 1-31.
Pan, I 2016, Developing an orphan drug? Understand the FDA guidance, Web.
Schilling, M 2012, Strategic management of technological innovation, McGraw-Hill Education, New York, NY.
Toften, K & Hammervoll, T 2013, ‘Niche marketing research: status and challenges’, Marketing Intelligence & Planning, vol. 31, no. 3, pp. 272-285.