Introduction
The isolation and identification of microorganism such as bacteria constitute a systematic process that applies physical and chemical attributes in the laboratory environment. Given that microorganisms are diverse and ubiquitous, the isolation of a pathogenic species that caused the death of Drosophila melanogaster (fruit flies) is a critical step in the study. The isolation involves separating a particular strain or species of bacteria from diverse microorganisms that live in a certain environment (Madigan, Martinko, Bender, Buckley, & Stahl, 2015). Surface plating, selective media, incubation, and inoculation are common laboratory approaches used in the isolation of bacteria (Medina et al., 2017). Surface plating involves a series of streaks on plated media until pure colonies of bacteria emerge.
Selective media allows the isolation of bacteria using nutrients that enhance or antibiotics that suppress the growth of a particular strain. Incubation is an approach that uses atmospheric surroundings, such as temperature, aerobic condition, anaerobic state, and carbon dioxide concentration, in selecting bacteria that survive in a stringent environment. Inoculation of bacteria into appropriate host permits selection for microorganisms survive under specific physiological conditions. Phenotypic characteristics of colonies, such as shape, size, elevation, surface texture, edge patterns, colour, structure, the degree of growth, and the nature of spread, improve the isolation of pure colonies in the growth media. Thus, a combination of these approaches has enhanced the isolation of bacteria.
Following the isolation of pure colonies is the identification to establish where bacteria fall into the classification scheme. The common laboratory techniques employed in the identification of bacteria are the morphology of colonies and cells, biochemical reactions, physiological mechanisms, and genetic characteristics. The morphology of colonies includes size, shape, surface texture, colour, motility, and edge patterns among other features (Teeseling, Pedro, & Cava, 2017). Additionally, the morphology of cells can classify bacteria into five groups, namely, rod-shaped (bacilli), spherical-shaped (cocci), comma-shaped (vibrio), corkscrew-shaped (spirochaetes), and spiral-shaped (spirilla) (Constantino, Jabbarzadeh, Fu, & Bansil, 2016). In this case, the unknown bacterium was a bacillus for its morphology depicted it as rod-shaped.
Biochemical methods identify the physiological features of bacteria. These methods are staining, nitrate test, indole test, methyl red test, citrate test, catalase test, motility agar, ONPG test, ornithine decarboxylase test, xylose fermentation test, and mannitol test (Madigan et al., 2015). In this view, the experiment applied these approaches in the isolation and identification of the unknown bacteria. In an observation made in the laboratory, about 40% of fruit flies (D. melanogaster) used in a class experiment died due to infections caused by unknown microbes. The dead flies were examined, and pathogenic microbes in them were isolated for identification.
The following are two aims of the experiment performed in the laboratory to isolate and isolate the unknown bacteria:
- To isolate unknown pathogenic microbe that infected fruit flies and caused their death.
- To identify unknown pathogenic microbe using phenotypic features and biochemical tests.
Materials and Methods
(Incomplete, to be done by the customer)
The unknown bacteria were isolated from D. melanogaster (fruit flies), which died during a class experiment. The isolated bacteria were cultured under aerobic conditions in a nutrient agar plate for 24 hours at 350C. Morphological characteristics of the unknown bacteria were observed and a series of biochemical tests performed, leading to the identification.
Results
Results Part A
Table 1 below summaries the results of the experiment by showing biochemical tests performed, observations made, and interpretations.
Table 1: Tests and Observations that Identified the Unknown Bacterium.
Results Part B
Following culture and incubation, morphological characteristics were used to identify the unknown microbe. The observations made showed that the unknown microbe was a rod-shaped bacterium. Subsequently, a series of biochemical tests were sequentially performed and deductively interpreted, leading to the identification of the bacterium that caused the death of fruit flies as P. rettgeri. Gram staining results revealed that the unknown bacterium was Gram-negative because it retained Safranin stain instead of crystal violet stain. The analysis of the conditions of growth showed that the unknown bacterium was a non-fastidious aerobe since it grew in a simple nutrient agar in the presence of oxygen. Catalase test indicated that the unknown bacterium was an aerobe for its degraded hydrogen peroxide and generated visible oxygen bubbles. In determining the ability of the unknown bacterium to produce indophenol blue as the product of oxidising α-naphthol and dimethyl-p-phenylenediamine, oxidase test gave a negative test. The test implies that the unknown bacterium was unable to oxidise α-naphthol and dimethyl-p-phenylenediamine and produce indophenol blue. Therefore, morphological features, Gram staining, catalase test, and oxidase test confirmed that the unknown bacterium was a rod-shaped and Gram-negative aerobe with the ability to oxidise hydrogen peroxide but not indophenol blue.
As biochemical tests narrowed down to Acinetobacter and Enterobacteriaceae, nitrate reduction test was used to differentiate the two. Nitrate reduction test depicted a positive outcome of Enterobacteriaceae due to the ability of the bacterium to convert nitrate into nitrite, which combined with the sulphanilic acid to form a red-coloured complex of nitrite-sulphanilic acid. Motility test demonstrated that the unknown bacterium was a motile rod as depicted by the cloudiness of the media. Motile bacteria clouded the media because they moved away from the inoculated regions using flagella or cilia. Examples of motile bacteria are Escherichia, Salmonella, Enterobacter, Proteus, Morganella, Serratia, Citrobacter, and Providencia. Subsequently, the indole test was used to identify bacteria that formed the indole ring owing to the presence of tryptophanase. The indole test showed that Escherichia, Morganella, Proteus, and Providencia were positive for tryptophanase, whereas Salmonella, Enterobacter, Serratia, Proteus, and Citrobacter were negative for tryptophanase.
The urease test ascertained the ability of the bacterium to convert urea into carbon dioxide, ammonia, and water. Ammonia produced created an alkaline environment that turned phenol red into deep pink, which was a positive indicator for the presence of urease. MacConkey agar revealed that the bacterium was unable to ferment lactose, while oxidative-fermentation of glucose showed that the bacterium fermented glucose. Further biochemical analysis using the ONPG test identified bacteria with or without beta-galactosidase. Bacteria with beta-galactosidase hydrolysed ortho-Nitrophenyl-β-galactoside into ortho-nitrophenol and galactose with the former product forming a yellow-coloured compound. Escherichia has a positive ONPG test, while Morganella, Providencia, and Proteus have a negative ONPG test. Xylose test gave a negative outcome, which showed that the bacterium was Morganella and Providencia for it did not produce acids from xylose.
Further identification using ornithine decarboxylase test indicated that the bacterium utilised glucose and generated its acids, which reduced pH and caused bromcresol to turn from purple to yellow. Due to the lack of ornithine decarboxylase, the bacterium showed a negative test for it failed to change the observed colour from yellow to purple. In this view, the persistence of yellow colour indicated that the bacterium was Providencia. The use of methyl red confirmed that the bacterium produced acids from glucose through mixed acid fermentation. Citrate utilisation test revealed that the bacterium exhibited the activity of citrate lyase and oxaloacetate decarboxylase because colour changed into blue. Voges Proskauer test showed negative outcome as the bacteria produced mixed acids and acetoin, the major products of glucose metabolism. Mannitol fermentation test eventually identified the bacterium as P. rettgeri because it was able to utilise mannitol as a source of carbon and produced acids that turned phenol red yellow.
Discussion
Phenotypic characterisation of the unknown bacteria formed the basis for biochemical tests and subsequent identification. At the commencement of the identification process, the unknown bacteria appeared as rods in their shape. Constantino et al. (2016) argue that the shape is an adaptive feature because rod-shaped bacteria are more mobile than spherical-shaped bacteria, and thus they easily respond to chemotactic stimuli appropriately. The first chemical test, Gram stain test, identified the unknown bacteria as Gram-negative. Gram staining classifies bacteria into two main categories, namely, Gram-negative and Gram-positive, depending on the properties of their cell wall. Gram-negative bacteria do not retain Gram stain because they have a thin layer of peptidoglycan, whereas Gram-positive bacteria retain Gram stain because they have a thick layer of peptidoglycan (Becerra, Roy, Sanchez, Christy, & Burmeister, 2016). As the bacterium grew in a simple nutrient agar in the presence of oxygen, it exhibited features of a non-fastidious aerobe.
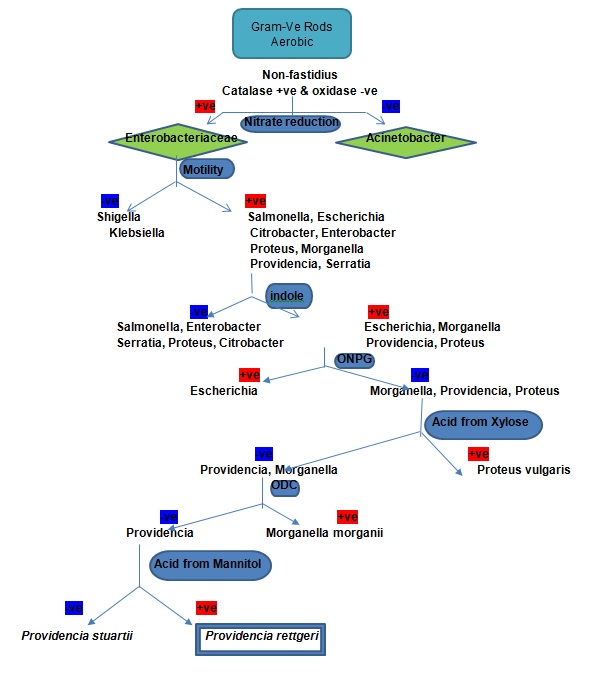
Chemical tests confirmed that the bacteria belonged to the family of Enterobacteriaceae due to positive catalase test, negative oxidase test, and positive nitrate reduction test. Observed bubbles in catalase test indicated that the bacteria produced oxygen when it metabolised hydrogen peroxide placed in the media. Madigan et al. (2015) explain that a bacterium with a positive test produces catalase enzyme, which catabolises harmful hydrogen peroxide into harmless products of oxygen and water. Negative oxidative test means that the bacterium lacked cytochrome c oxidase to convert tetramethyl-p-phenylenediamine into a purple-coloured product called indophenol. In essence, the bacterium exhibited properties of aerobic, anaerobic, and facultative bacteria.
A positive nitrate reduction test implies that a bacterium can undertake the nitrification process by breading down nitrates into nitrite and nitrogen (Madigan et al., 2015). According to Mardaneh and Dallal (2016), nitrate reductase test differentiates bacteria in the family of Enterobacteriaceae from those in other families, which do not catabolise nitrate into nitrite. Motility test indicated that the unknown bacterium was one of the motile bacteria, namely, Escherichia, Salmonella, Enterobacter, Proteus, Morganella, Serratia, Citrobacter, and Providencia, in the Enterobacteriaceae family. Constantino et al. (2016) explain that rod-shaped bacteria have flagella and streamlined body enables them to swim in their environment. The media became cloudy because the bacterium left trails on the media during migration using cilia and flagella.
Further differentiation of the unknown bacterium using the indole test showed that it could be Escherichia, Morganella, Proteus, and Providencia. Bacteria that form indole ring have tryptophanase activity, which breaks down tryptophan into indole and pyruvate (Islam, Akbar, Akther, & Islam, 2016). The urease test and oxidative-fermentation of glucose showed that the bacterium could catabolise urea and ferment glucose respectively. MacConkey agar and the ONPG test revealed that the bacterium was a non-lactose fermenter. Morganella, Providencia, and Proteus are three genera of bacteria that exhibit a negative ONPG test (Jemilehin, Ogunleye, Okunlade, & Ajuwape, 2016). The ONPG test eliminated Escherichia and narrowed down to the three genera of bacteria.
Negative tests of xylose and ornithine decarboxylase indicated that the unknown bacterium belonged to the genus of Providencia. As the bacterium lacked ornithine decarboxylase, it did not breakdown ornithine. Citrate utilisation test, methyl red, and mannitol test confirmed that the bacterium carried out fermentation and generated acids. Citrate lyase and oxaloacetate decarboxylase are enzymes that degrade citrate and oxaloacetate to produce diethyl, ethanol, and acetic acids (Chen et al., 2017). However, a negative Voges Proskauer test implied that the bacterium was unable to convert diethyl into acetoin (Chen et al., 2017). Eventually, mannitol fermentation test indicated that the bacterium was P. rettgeri because it metabolised mannitol.
The analysis of the experiment shows that it attained its aims of isolating and identifying the unknown bacterium that made fruit flies die. Since bacteria are diverse and ubiquitous, successful isolation of the unknown bacteria formed the basis of its identification as a rod-shaped strain. Moreover, consecutive chemical tests successfully identified the unknown bacteria through the elimination method. Outcomes of diverse chemical tests corroborated each other, resulting in the accurate and valid identification of unknown bacteria (Monteiro et al., 2016). Therefore, the experiment achieved its main objectives for it managed to isolate and identify the unknown bacterium up to the species level as P. rettgeri.
The major problem encountered during the isolation and identification of the unknown bacterium was contamination of cultures by other microbes. Given that bacteria are ubiquitous, preparation of cultures was prone to contamination. Jain, Aravindaram, and Pal (2016) explain that contamination of cultures by other microbes impedes isolation and identification of bacteria in the laboratory. Another problem encountered was confusion in the performance of biochemical tests and interpretation to give accurate results. Preparation of broth, incubation of bacteria, and observation of different colours were prone to errors, which influence ultimate outcomes of biochemical tests. Since P. rettgeri does not decompose sulphur-containing amino acids, the use of hydrogen sulphide tests, such as Kligler iron agar, sulphite indole motility, and lead acetate paper tests, are useful in the identification of the unknown bacterium.
References
Becerra, S. C., Roy, D. C., Sanchez, C. J., Christy, R. J., & Burmeister, D. M. (2016). An optimised staining technique for the detection of Gram-positive and Gram-negative bacteria within tissue. BMC Research Notes, 9(1), 1-10. Web.
Chen, C., Zhao, S., Hao, G., Yu, G., Tian, H., & Zhao, G. (2017). Role of lactic acid bacteria on the yogurt flavor: A review. International Journal of Food Properties, 20(1), 316-330. Web.
Constantino, M. A., Jabbarzadeh, M., Fu, H. C., & Bansil, R. (2016). Helical and rod-shaped bacteria swim in helical trajectories with little additional propulsion from helical shape. Science Advances, 2(11), 1-14. Web.
Islam, K. N., Akbar, T., Akther, F., & Islam, N. N. (2016). Characterisation and confirmation of Lactobacillus spp. from selective regional yoghurts for probiotic and interference with pathogenic bacterial growth. Asian Journal of Biological Sciences, 9(1), 1-9. Web.
Jain, A., Aravindaram, K., & Pal, D. (2016). Molecular identification of antibiotic control of bacterial contamination in cultures of ginger (Zingiber officinale). The Journal of Horticultural Science and Biotechnology, 19(2), 122-128. Web.
Jemilehin, F. O., Ogunleye, A. O., Okunlade, A. O., & Ajuwape, A. T. P. (2016). Isolation of Salmonella species and some other Gram-negative bacteria from rats cohabiting with poultry in Ibadan, Oyo State, Nigeria. African Journal of Microbiology Research, 10(29), 1104-1110. Web.
Madigan, M., Martinko, J., Bender, K., Buckley, D., & Stahl, D. (2015). Brock biology of microorganisms (14th Ed.). New York, NY: Pearson Education.
Mardaneh, J., & Dallal, M. M. S. (2016). Isolation and identification of Enterobacter asburiae from consumed powdered infant formula milk (PIF) in the neonatal intensive care unit (NICU). Acta Medica Iranica, 54(1), 39-43.
Medina, D., Walke, J. B., Gajewski, Z., Becker, M. H., Swartwout, M. C., & Belden, L. K. (2017). Culture media and individual hosts affect the recovery of culturable bacterial diversity from Amphibian skin. Frontiers in Microbiology, 8(1574), 1-14. Web.
Monteiro, A. C. M., Fortaleza, C. M. C. B., Ferreira, A. M., Cavalcante, R. S., Mondelli, A. L., Bagagli, E., … Cunha, M. L. R. (2016). Comparison of methods for the identification of microorganisms isolated from blood cultures. Annals of Clinical Microbiology and Antimicrobials, 15(45), 1-11. Web.
Teeseling, M. C. F., Pedro, M. A., & Cava, F. (2017). Determinants of bacterial morphology: From fundamentals to possibilities for antimicrobial targeting. Frontiers in Microbiology, 8(1264), 1-17. Web.