Overview of XRF
XRF (X-ray fluorescence) is an analytical technique that verifies the chemical elements found in a material without destroying it during analysis (Pinto 2018). Specialised equipment known as XRF analysers establish a sample’s chemistry by quantifying the extent of fluorescence emitted following the excitation of the material by X-ray radiations. During XRF, a sample (can be in solid or liquid form) is exposed to high-energy X-rays originating from a regulated X-ray tube.
When radiations possessing energy that exceeds the binding energy of atomic K or L shells hit the atoms in the material, an electron from the innermost orbital shells is displaced, which destabilises the atom (Leyden 2018).
As the atom returns to stability, the void left by the excited electron is filled with another electron from an orbital with higher energy. Fluorescent X-rays are emitted during this transition. Therefore, the irradiation of a chemical element with X-rays generates a set of distinct fluorescent X-rays that are unique to a given element. This array of fluorescence is referred to as a fingerprint and can be used to identify various chemical elements (Klockenkämper & Von Bohlen 2014). As a result, XRF spectroscopy can be used to conduct quantitative and qualitative investigations of material composition (Rouillon & Taylor 2016).
Experimental Procedure
About 20 unknown, numbered salt samples were provided to the class. One unknown sample (number 16) was chosen and used for individual analyses. An XRF film was placed over one part of a cup. A second part of the cup was then put on top of the film and used to push it down. A weighing balance was cleaned to remove any impurities that could have altered the accuracy of the measurements after which approximately 2.48 grams of the unknown sample were weighed.
The required weight was supposed to be between 2 and 3 grams. The weighed salt sample was then put in a sample press machine to be flattened after which the height of the flattened sample was measured and found to be 4 cm. The sample was then labelled by indicating the sample number, weight, name of the student and height after flattening. These values were indicated on the cover of the cup. The prepared sample was then put in the RIGAKU XRF machine for analysis together with additional unknown samples prepared by other students. The results of the analyses were later provided by the instructor to facilitate the writing of the report.
Results
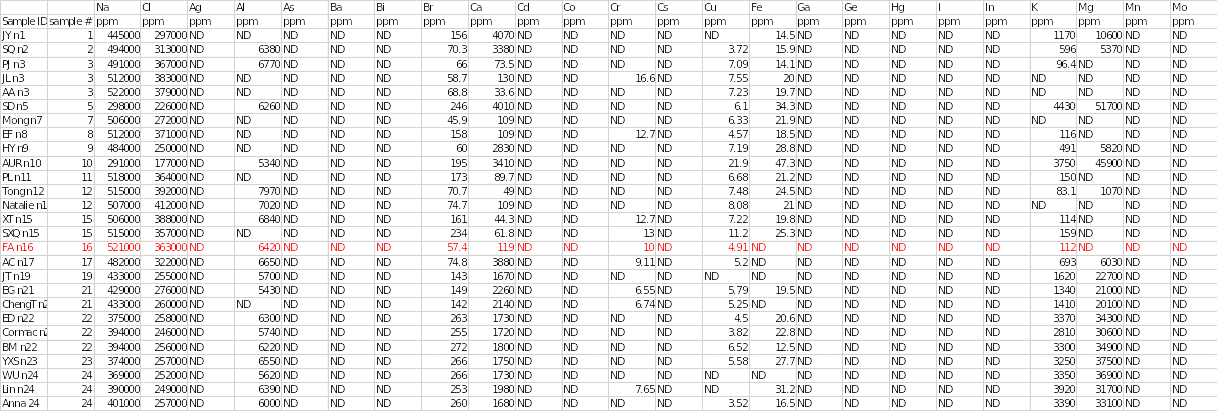
Table 1 below shows the concentrations of various trace elements in different salt samples. The individual sample is highlighted in red. It was noted that sodium (Na) was the main cation with a concentration of 521,000 parts per million (ppm), whereas chloride (Cl) was the predominant cation with a concentration of 363,000 ppm. Sample 16 had the second highest concentration of sodium.
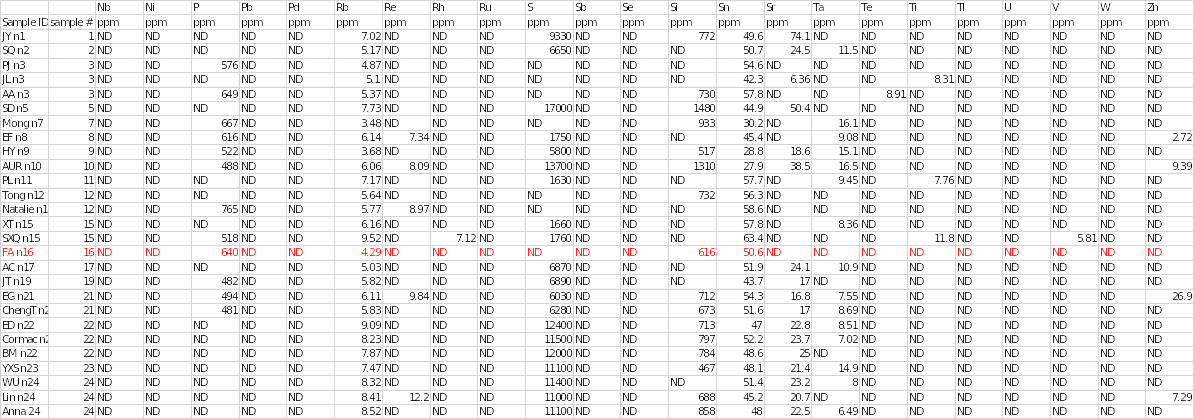
Trace elements that were present in sample 16 included aluminium at 6420 ppm, bromine at 57.4 ppm, calcium at 119 ppm, chromium at 10 ppm, copper at 4.91 ppm and potassium at 112 ppm. Other chemical elements that were found in the salt specimen included phosphorous, rubidium, silicon and stannum at concentrations of 640, 4.29, 616 and 50.6, respectively. Trace elements that were detected in other samples but were absent in sample 16 included iron, magnesium, rhodium, sulphur, strontium, tantalum, titanium, tellurium, vanadium and zinc.
In contrast, some elements were not completely detected in any of the salt samples. They included silver, arsenic, barium, bismuth, cobalt, caesium, gallium, germanium, mercury, iodine, indium, manganese, molybdenum, niobium, nickel, lead, palladium, ruthenium, antimony (stibium), selenium, thallium, uranium and tungsten. Sodium and chloride were the most abundant elements in all salt samples.
Discussion and Conclusion
Based on the XRF findings of the salt specimen, the unknown salt sample was likely to be table salt. The unidentified sample was pure white with very fine crystals, an observation that was in line with the appearance of table salt (Anchell 2016). The specimen also contained large quantities of sodium and chlorine, which is usual for table salt (De la Guardia & Garrigues 2015). The sample could not be sea salt because it lacked sulphur, which makes up sulphates that are found in sea salt. Trace minerals that can be found in sea salt include bromine, phosphorus, boron, iron, zinc, copper, manganese and silicon (Ohji et al. 2017). However, the unknown analyte did not contain constituents such as zinc and manganese, which are among the most common trace elements in sea salt (Kadko, Landing & Shelley 2015).
The sample could not be rock salt because it did not contain barium and strontium. Studies show that rock salt contains very large quantities of these two elements (Kelly, Findlay &Harris, 2018). Therefore, their absence in the specimen ruled out the possibility of rock salt. The findings of this study show that XRF is a very reliable analytical technique in the determination of the elemental composition and concentration of chemical substances.
Reference List
Anchell, S 2016, The darkroom cookbook, 4th edn, Routledge, New York, NY.
De la Guardia, M & Garrigues, S 2015, ‘Handbook of mineral elements in food, John Wiley & Sons, Hoboken, NJ.
Kadko, D, Landing, WM & Shelley, RU 2015, ‘A novel tracer technique to quantify the atmospheric flux of trace elements to remote ocean regions’, Journal of Geophysical Research: Oceans, vol. 120, no. 2, pp. 848-858.
Kelly, VR, Findlay, SE & Harris, C 2018, ‘Chemical composition of rock salt brine compared with brine from oil and gas wells’, Journal of Environmental Engineering, vol. 144, no. 9, p. 06018006.
Klockenkämper, R & Von Bohlen, A 2014, Total-reflection X-ray fluorescence analysis and related methods, John Wiley & Sons, Hoboken, NJ.
Rouillon, M & Taylor, MP 2016, ‘Can field portable X-ray fluorescence (pXRF) produce high quality data for application in environmental contamination research?’, Environmental Pollution, vol. 214, pp. 255-264.
Ohji, T, Colorado, H, Matyáš, J & Kanakala, R 2017, Advances in materials science for environmental and energy technologies vi, volume 26, John Wiley & Sons, Hoboken, NJ.
Pinto, AH 2018. ‘Portable X-ray fluorescence spectrometry: principles and applications for analysis of mineralogical and environmental materials’, Aspects in Mining and Mineral Science, vol. 1, pp. 1-6.