Introduction
A lubricant’s de-waxing process involves removal of paraffinic hydrocarbons so as to improve the pour point of the feed stock. The pour point of oil is the lowest temperature in which oil can flow, pour or move when refrigerated or cooled without disruption under certain fixed conditions.
De-waxing produces high yields of products of improved quality base oils. Waxes require isomerization for their conversion into lubricant base oils. De-waxing processes create pure un-branched hydrocarbon chains without either aromatic contents or unwanted hetero-atoms in gas to liquid (GTL) processes (Gary &Handwerk, 2001).During the de-waxing process, the solvent is subjected to a mixture of the waxy oil.
In the catalytic de-waxing mechanism, wax components are reduced in the reaction to recapture the de-waxed oil molecules. A de-waxing process involves prior solvent extraction before the process is done. Heavy gas and oil from a refinery that is usually at very high temperatures is usually the feed stream. The gas is usually at high temperatures of 95 degrees Fahrenheit. This fact allows the feedstock not to crystallize.
Shock treatment can cause formation of small crystals by the wax which would block the filter cloth during filtration process hence lowing wax recovery (Speight, 2010). The solvent is usually of equal temperature with the wax stream. It is then filtered through rotary filters. The primary rotary filter separates the stream into de-waxed oil or wax solvent stream. The de-waxed oil stream undergoes heat integration and continues to solvent recovery process.
The wax or oil stream proceeds to the secondary filter. Here, it is separated into mostly solvent and wax with 3wt% oil content. The oily wax is de-oiled by re-melting to release trapped oil. The final filtering step is the tertiary filter. It separates waxes into hard and soft ones. Soft wax is recycled and hard one is re-slurred and purified to food grade wax.
De-waxing products can be categorized into two:
- Base oil which is used to make lube oil for automotive and industrial lubricants and for production of automatic transmission fluids.
- Food grade wax which is used to produce sealants. Crayons, cosmetics, foods and candles are also produced from this wax.
Lubricants’ oil de-waxing methods
Lubricant manufacturers may use either catalytic or urea de-waxing or solvent waxing. During the catalytic de-waxing process, there is usually selective removal of lighter non- normal hydrocarbons and also an even removal of normal paraffin. Urea de-waxing is normally used to produce low pour points using urea.
This process of de-waxing is good for recovering refrigeration and transformer oils because no refrigeration is required. It is important to include a desirable solvent because urea has no MEK properties. The viscosity of the oil has negative results in its contact between oil and urea. Difficulties are encountered during filtration, hence the addition of solvents.
MSDW’s process
This process uses a two-stage cascade system. It is shaped like a selective de-waxing catalyst in the second stage (Fleig, 2005). This system is highly flexible and functions with hydrogen pressures between 27 to 207 bars. The waxy oil and the hydrogen are mixed and then heated. They are then directed to the top of the de-waxing reactor. Downstream, the by-products which are basically low sulfur distillate, high-octane, naphtha and hydrogen-rich recycled gas are separated from the lube base oil (Pujado &Jones, 2006).
Propylene- acetone’s de-waxing process
Propylene and acetone is the solvent mixture in this process. This process is adaptable to de-waxing plants that utilize propane instead of MEK (Fahim, Al-Shhaf&Elkilani, 2009).Crystallization as a process is complex as it is usually determined by the rate of cooling of crystals. High and low cooling has an impact of the speed of the crystallization process and the quality of formed crystals. Propane is widely available as a byproduct in refineries and therefore its utilization is easier.
This process leads to production of elements with low pour points, low refrigeration loads and reduced filter usages. Industrial technological research should focus on creation of catalysts that enhance the de-waxing of propylene and acetone instead of overreliance on products like propane. This fact would ensure that the performance of the solvent raw materials in this process is enhanced.
MEK de-waxing method
Methyl Ethyl Ketone (MEK) is an anti-oil solvent and thus dissolves little wax at low temperatures. It is a wax precipitating agent. Toluene is an oil solvent that dissolves the oil. At low temperatures, it maintains fluidity.
A combination of major solvents is used in solvent de-waxing. MEK has become the most commonly used anti solvent in modern times but over reliance on it could lead to possible depletion of raw materials used in making it and consequent production of counterfeit products in the market. Industrial experts have advised that more research should be done to create new anti-oil solvents to complement the use of MEK.
Nature of MEK de-waxing
MEK has a poor solvent power and selectivity of paraffinic elements. It precipitates the wax leaving the de-waxed base oil. Studies have shown that the combination of toluene and MEK solvents in de-waxing processes causes an optimum wax precipitation of 40-75 v %. With increased MEK, the oil’s viscosity index increases but the solid point is maintained.
Process of MEK
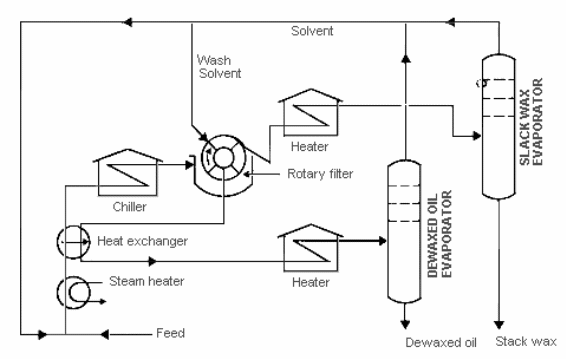
The solvents used in this process are MEK and toluene which are mixed with the wax or oil stream feed. The feedstock is usually of high temperature; about 95°Farenheit therefore, the solvent added should have corresponding temperature. The solvent of the same temperature is again added.
This fact prevents shock treatment. The mixture is then filtered through the rotary filters which separate the stream into de-waxed oil or wax solvent stream.
Process variables
MEK is highly preferred because of its distinct anti-oil solvent characteristic. All the other solvents have almost similar properties but MEK or toluene combination is the most commonly used one in refineries. High solvent as opposed to oil use improves the process of filtration. A high solvent facilitates faster crystal growth reducing the viscous nature of the varied streams. At 100 wt. % MEK, there is a significant wax recovery. High cooling rates have been reported to form undesirable wax that is hard to filter since it blocks the filters.
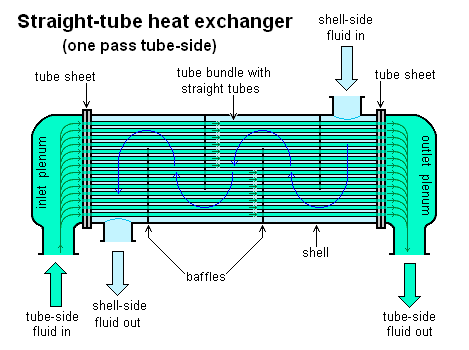
Surface pressure exchanger
The heat exchanger is used for crystallization of the wax. The process involves the use of the mid of the heat exchanger, the heated stream flows inside as the coolant flows on the outside. Wax crystals form slowly as the stream cools (Prestoris, 2004).
The fact shows that there is usually poor heat conduction between the waxy feed and the coolant due to the formation of wax crystals. The surface heat exchanger contains scraper blades which are usually inconstant rotation (Gary et al, 2007). Their rotation removes clogged wax inside the pipe. This fact allows formation of more wax crystals.
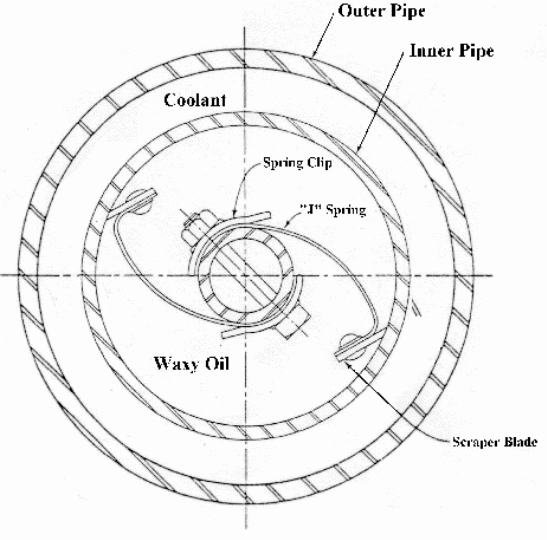
Vacuum rotary filter
Vacuum rotary filters are drums with a vacuum inside them covered by cloth. This fact prevents entry of the wax into the drum as the oil seeps in. This process removes any oil that might have been left in the wax. As the drum rotates, the scrapers remove the wax. It is deposited into a collection basin where it is re-slurred and re-melted with more solvent. It is used in separation of aromatic impurities and naphthalene by precipitation or dissolving of the components.
Filters’ process operation
In its operation; liquid is sent to tube units set below a drum. The drum cycles through the liquid and the vacuum pulls the solids away from the liquid into the drum pre-coat system. The liquid sucked through the filter causes the solid to stick to the outer surface of the drum.
Maintenance and protection of the system
Maintenance and protection of the system has to involve the maintenance of the various filters. The oil or the coolant filters has to be changed after every 500 hours so that they can clean away the contaminants. The oil separator has to be changed with change in oil for it to separate oil from air effectively. The drive belt has to be changed after 800 hours for effective performance.
Other equipments used in this process include chillers with heat exchangers which gradually lower the temperature of the mixed stream of solvent and wax or oil to allow crystallization to take place. The others are the de-waxed oil and slack wax evaporators.
Features of the vacuum rotary filters
Their features include drum, valve, drum deck, filter cloth, internal piping, agitator and the tank. These features are small in size and structure to facilitate faster operation. They are powerful and suffer low labor intensity making them easy to operate. The drum is supported by the trunnion which is set at the end of valve. It helps in formation of the vacuum cell at the bearing end. The valves are used to regulate the cycle sequence.
Each portion is exposed to the vacuum, the dead zone and the blow. The valve contains adjustments blocks and fixed forms which creates the drying ratio in the filtration process. The internal piping can be single or double row piping to facilitate the channeling of the filtrates, steam and waste.
The drum deck is separated into compartments waved to the vacuum or the rotating drum. The filter cloth acts as the cake tied to the drum face and it’s made from propylene or polyester. The agitator suspends the sludge material as the tank houses the drum and the agitator.

Vacuum and rotary filters’ process variables
The process variables in rotary filtration involve temperature, pressure, solid content and particle size and distribution (Parkas, 2003). The variables define the present status of the process. The temperature is usually maintained high with operational temperatures going as high as 2000C. High temperatures are required for distillation, evaporation and thermal breakdown.
Pressure is crucial and is maintained at 6 bars with the pressure filters taking different timing like 10 minutes for cake application and 8 hours for polishing. The solid contents take about 50% or more of the weight with the particle distribution being 1 to 2 microtones.
Chillers
These equipments are heat exchangers which gradually lower the temperature of the mixed stream of solvent and wax or oil to allow crystallization to take place (Lum, 2011). They are specially designed to scrap out the wax deposits in the heat exchanger surface so as to have efficient heat transfer. Chilling involves direct heat transfer.
Evaporators
The evaporator is used in recovery of the solvent from the de-waxed oil and wax. Components include the motor which rotates the evaporating flask, vapor duct that channels the sample, vacuum system that reduces pressure, water bath for heating the sample and the condenser that acts as a coolant.
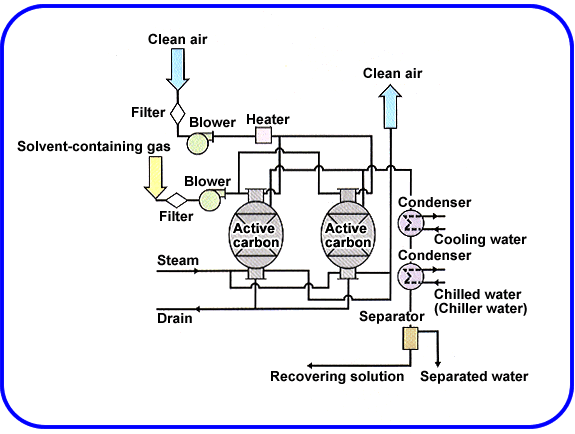
Solvent recovery system
The solvent recovery system supplies parts and services for recycling equipment, wastewater treatment and washing of parts. The process reduces the cost of disposing solvents and waste water charges. The solvent recovery system involves the following process as shown in the diagram.
Solvent used
The solvent de-waxing stem involves MEK, toluene, propane benzene ,methylene chloride, ethylene dichloride, sulfur dioxide and cyclohexane.MEK is used in industries in the dissolution of wax at low temperatures.
A graphical comparison of the various solvents as per the table is as follows
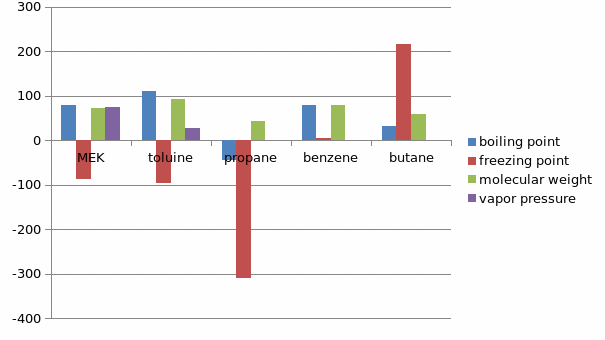
Table : 24.2
A new development in lubricants’ de-waxing processes
The industry is facing a lot of development with respect to technological advancement. Nowadays, there is greater use of the skill in solvent extraction. Industries are producing edible vegetable oil from oil seeds and protein units that are nutritious and economically viable. Capital investment is prioritized.
The industry is making efforts in investment and money making through incorporation of various ideas in respect to this industry. The food industry also applies the PEG-NaCl system to fragment small molecules like nucleic acids and peptides. Information on DNA purification is important in generating biotechnological purified genetically material.
Conclusion
The MEK lubricant and de-waxing method have proven to be useful industrial components (Favennec, 2001). More research should be done to improve their performance in industrial processes. The government should allocate more funds to safeguard industrialists from fraudulent people who sell counterfeit products hence reducing the marketability of the MEK lubricant.
The de-waxing method has also proved to be a resourceful one and scholars should devise easier ways of understanding the solvent extraction system, heat exchanger and the functioning of the filters because it is important considering that health and ethical standards have to be adhered to.
References
Fahim, A.M., Al-Shhaf, A.T&Elkilani, A. (2009). Fundamentals of petroleum refining. Amsterdam, Netherlands: Elsevier Science.
Favennec, J.P. (2001). Petroleum Refining, Refinery operation and management. Paris, France: Editions Technip.
Fleig, F. (2005). Oil Empire: Visions of prosperity in Austrian Galicia (Harvard Historical Studies. Harvard, United Kingdom: Harvard University Press.
Gary, J. et al. (2007). Petroleum refining: technology and economic. New York, USA: CRC Press.
Gary, H.J., &Handwerk, E.G. (2001). Petroleum Refining. New York City, USA: Routledge Taylor & Francis.
Lum, G. (2011). The negotiation field book. Simple strategies to help one negotiate everything. New York City, USA: McGraw-Hill companies.
Parkas, S. (2003). Refining process hand book. Burlington, USA: Elsevier Science.
Prestoris, A. (2004). Compositional Analysis of Naphtha and Reformats. New York, USA: CRC Press.
Pujado, R.P., &Jones, J.S. (2006). Handbook of petroleum. Rotterdam, Netherlands: Springer.
Speight, G.J. (2010). The refinery of future. Oxford, United Kingdom: Elsevier Science.