Hereditary Fructose Intolerance
According to Wendell W. Weber, Fructose and Lactose are the two main sources of glucose for the human body. These complex sugar molecules are broken down into glucose which serves to be an energy source to be used by various body organs and systems to accomplish or perform various activities. (Weber, n.d., p. 234).
As in case of SH (patient), the symptoms are seen or become severe with the intake of fructose containing fruit juice or baby food. Also the general symptoms (pale skin and eyes, non-functioning liver and kidney), tremors, decreasing appetite, slow growth etc. have been related to fructose intolerance i.e. the body of the infant has stopped responding to or treating fructose in the desired way. The fructose being consumed through food is not being properly metabolised and thus accumulation of it along with the lack of glucose (generated upon metabolism of fructose) is the cause of above mentioned physically noticeable symptoms. Occurrence of Fructose Intolerance can be related to genetic or hereditary factors. The single gene for the intolerance of fructose (mutated gene for production of enzyme ALDOB) might be present as a recessive gene (single copy present) in each parent. In the children of such carrier parents, both the copies of this recessive gene occur together; the recessive gene exhibits its character and results in Hereditary Fructose Intolerance. ALDOB is the most important enzyme used in the metabolic pathway of fructose or other complex sugar molecules. The enzyme is also referred to as, Aldolase B or Fructose Aldolase B (Hereditary Fructose Intolerance, 2011).
Following diagram depicts the essential steps involved in fructose breakdown in the body and the enzymes associated with each step:
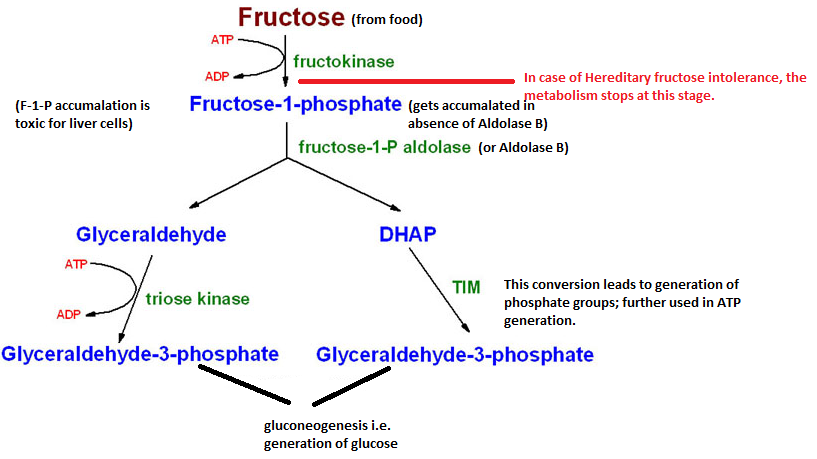
From the above diagram, it is known that the first most essential enzyme in the fructose breakdown cycle is Aldolase B or Fructose-1-P Aldolase. With improper or non-functioning of this enzyme Fructose-1-P does not get broken down to Glyceraldehyde and Dihydroxyacetone Phosphate (King, 1996). Accumulation of Fructose-1-P is itself toxic for the liver cells (leading to liver failure and jaundice), wherein the fructose metabolism indirectly causes Aldolase B deficiency (in case of SH, resulting from mutated recessive gene ALDOB obtained from parents) in the metabolic reaction on the by-products, Glyceraldehyde-3-Phosphate and phosphate groups, which are used in glucose (gluconeogenesis) and ATP production to fulfil the energy requirements of the body (Hereditary Fructose Intolerance, 2011). In the absence of gluconeogenesis, the patient is observed to have hypoglycaemia and retarded growth.
According to Lehninger, the main enzyme present in the liver is Glucokinase which metabolises glucose to generate energy. For utilizing fructose, aldolase B is thus necessary, which releases GHP molecules that are converted to glucose through gluconeogenesis cycle; and then acted upon by Glucokinase to give energy. The main substrate of the enzyme Fructose B or Fructose-1-P Aldolase is Fructose-1-Phosphate (Lehninger et al. n.d.). Thus, the main substrate for aldolase B is Fructose-1-Phosphate. The reaction between this enzyme and substrate is guided by their complementary structural configurations. Therefore the reaction between the two can be explained with the help of Emil Fisher’s Lock and Key Hypothesis as follows:
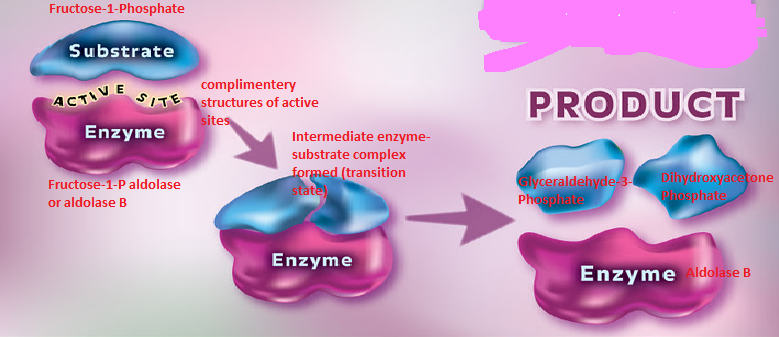
The above diagram elaborates the binding of enzyme (aldolase B) and the substrate (Fructose-1-Phosphate) at the active site of the enzyme. The structure of the active sites and the binding site of the enzyme and the substrate complement each other and thus can bind with each other only and not any other substrate or enzyme molecule (King, 1996). After the enzyme binds to its specific substrate (Fructose-1-Phosphate in this case), a temporary enzyme-substrate complex is formed which further breaks down to the products and the enzyme. Here the substrate undergoes a chemical change (forming products) but the enzyme gets released without any alterations in structure or function.
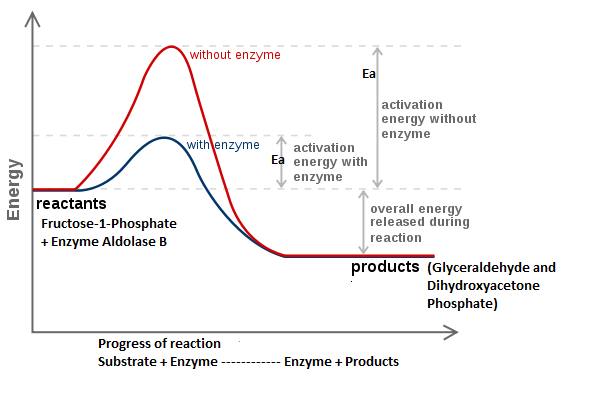
Activation energy (Ea) involved in the reaction (conversion of substrate to product) is the key factor which gets affected by the enzyme. Usually, the Ea required for a reaction is high and when an enzyme binds to the substrate the Ea level falls or reduces and the rate of reaction thus increases. In case of fructose metabolism also, the Ea required for forming Glyceraldehyde and DHD is high; but Aldolase B by combining with the fructose molecule effectively reduces the Ea level and thus the breakdown reaction occurs quickly and easily. Thus, an enzyme here acts as a catalyst which helps improve the rate of a biological reaction. Following graphs depict the effect of enzyme Aldolase B on the amount of Ea required for breakdown of fructose to Glyceraldehyde and DHD.
Mitochondrial Disease
Glycolysis is the breakdown of glucose and gluconeogenesis is the production of glucose molecules from different carbon sources. (Chapter 15: Cori and Alanine Cycles, n.d., p. 1). Glucose is the most important energy source required to maintain appropriate working state of organs and body systems. Any activity done by any organ or muscle requires energy which comes from breakdown of glucose molecules (glycolysis) to release ATPs (the energy currency). Liver is the main site of this energy producing glycolytic reaction apart from kidney and skeletal muscles where only small amount of glycolysis occurs. For all the above, glucose is the main starting substrate which is provided in the form of complex sugar molecules in various food items we consume. Like lactose, Galactose and sucrose also undergo specific enzyme-catalysed breakdown to release glucose. Apart from this, amino acids alanine and glutamate can also be used to as precursors for glucose formation reactions. Accordingly, all the conversions taking place at different sites (cells) that have been diligently linked with each other so that the products of one immediately get supplied as precursor for other reaction at another site (Ophardt, n.d.). As explained by Boron, one such cyclic process is Cori Cycle, named after its researcher. This cycle involves two types of cells; liver/hepatic and muscle cells.
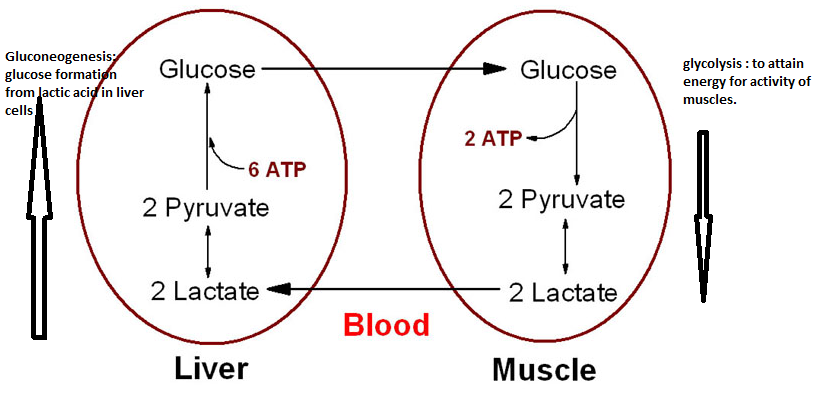
Here glucose is used to release ATP and lactate/lactic acid. This ATP is used as energy source by muscles and the lactic acid gets transferred to blood. Blood carries this lactic acid to liver where with the help of gluconeogenesis enzymes, this lactic acid gets combined with pyruvate and finally forms glucose molecules which again get utilised by muscle cells (in case of muscle activity) Thus, half part of Cori cycle occurs in muscle cells and another half takes place in the hepatic cells. (Boron & Boulpaep, n.d.).
In mitochondrial disease, as has been detected in the patient MW; if this inter-conversion from glucose to lactate and again to glucose is restricted to just one cell the process would be highly disturbed. If the interconversion gets restricted to muscle cell, the accumulation of lactic acid will lead to pain and cramps in the muscles. Also this will result in deficiency of lactic acid which acts as the carbon source for production of glucose resulting in less glucose, less ATP and less energy. Alternatively, if the cycle gets confined to only the hepatic/liver cell; no more glucose gets transferred to blood so firstly, the blood glucose level will fall and secondly muscle cells won’t get enough glucose to use as energy source leading to muscle fatigue.
According to Nelson and Cox, during aerobic conditions, citric acid cycle indirectly serves as the source of ATPs. This indirect but important role of citric acid cycle can be explained on the basis of its link with the electron transfer chain. The CO2 molecules produced during the citric acid cycle are necessary for initiation of the electron transfer chain and thus result in production of many ATPs. Also the main product of aerobic glycolysis (pyruvate) is consumed in the citric acid cycle only. Thus the citric acid cycle plays central role in aerobic respiration (using by products of glycolisis and producing precursors for electron transfer chain) (Lehninger et al. n.d.).
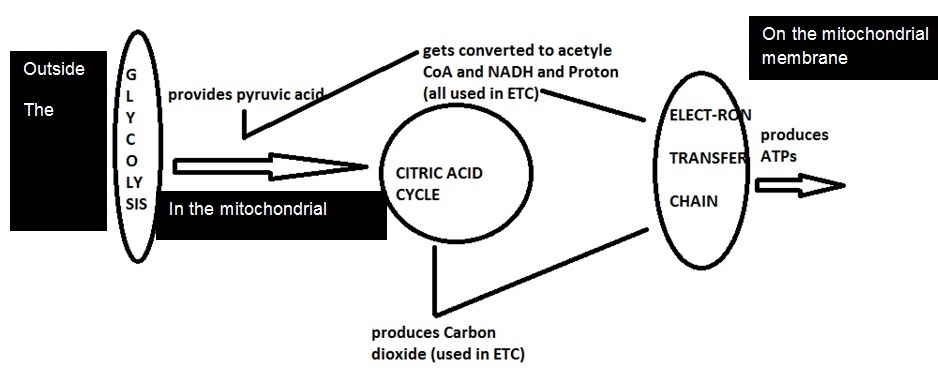
From the above model, it can be seen how citric acid cycle is centrally linked to aerobic glycolysis and electron transfer chain. The CO2 and protons produced during citric acid cycle are necessary for ETC and thus for ATP production. Mitochondrial membrane is the main site where electron transfer chain occurs. The process helps to build several ATP molecules which serve as energy sources in the body. Glycolysis outside the cell produces pyruvic acid, 2 ATPs, 2 NADH and H+. The NADH molecules and proton get involved in ETC to produce 6 or more ATPs and the pyruvic acid gets involved in the citric acid cycle (in the mitochondrial matrix), thus generating carbon dioxide molecules and protons which again get utilised in the ETC (on mitochondrial membrane) and release of ATPs. Coenzyme Q10 is the most essential and primary enzyme of the electron transfer chain which is required for synthesis of ATP molecules. When exercising; more glycolysis occurs in the muscle cells thereby producing more pyruvic acid and other by-products. These are rapidly converted to ATP with the help of citric acid and electron transfer chain on the mitochondrial membrane.
Now if as suspected in the above case study, a mitochondrial disease (mutation or defect in mitochondrial DNA) has occurred. It would mean the Coenzyme Q10 will not be produced. This depleted amount or deficiency of enzyme Q10 will inhibit the electron transfer chain at the last step where it is most required to convert ADP to ATP, thus resulting in energy deficiency and muscle fatigue. Therefore, observing the symptoms of our patient MW, it can be said that he might have had a mitochondrial disease and thus presenting with Hypotonia, motor weakness and fatigue.
Reference List
Boron, W. F. & Boulpaep, E. L. (n.d.). Medical Physiology: A Cellular and Molecular Approach. Hampshire: Prentice Hall.
Chapter 15: Cori and Alanine Cycles, (n.d.). 2011, Web.
Hereditary Fructose Intolerance. (2011). Genetics Home Reference. Web.
King, M. W. (1996). Fructose Metabolism. Themedicalbiochemistrypage.org. Web.
Lehninger et al. (n.d.). Principles of Biochemistry. Hampshire: Prentice Hall.
Ophardt, S. E. (n.d.). Cori Cycle. Virtual ChemBook: Elmhurst College. Web.
Weber, W. W. (n.d.). Pharmacogenetic. Oxford University Press, 234.