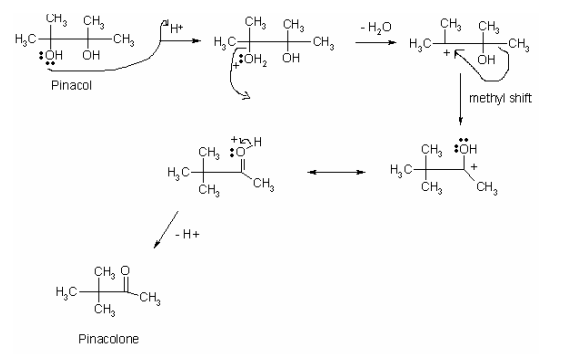
The pinacol rearrangement constitutes the dehydration of pinacol and the stabilization of carbocation by the shift of methyl. The mechanism of pinacol rearrangement follows the SN1 reaction mechanism with pinacol as a limiting reagent. The mechanism of the pinacol rearrangement commences with the protonation of one of the two –OH groups. Anslyn and Dougherty explain that the protons derived from the concentrated sulfuric acid get attracted to the lone pairs of electrons on one of the –OH group in the pinacol (675). The interaction of the proton with the –OH group leads to the formation of oxonium ion (-OH2+). The oxonium ion leaves the pinacol and creates a tertiary carbocation that causes a methyl shift in a manner that stabilizes it (Anslyn and Dougherty 675). The methyl shift culminates in the formation of pinacolone (ketone). Figure 1 below demonstrated the mechanism of pinacol rearrangement.
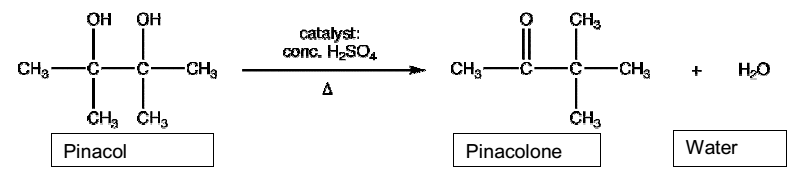
Evaluation of the experimental procedure shows that it was performed successfully as the pinacol rearrangement happened as expected. Comparative analysis of the theoretical yield and the actual yield of the experiment show a disparity. Starting with the 3 g of the pinacol, which is the limiting reagent, the theoretical yield of pinacolone could be 2.604 g. However, the actual yield obtained from 3 g of pinacol is 1.5 g, which is 57.6% of the theoretical yield. Therefore, the experiment managed to synthesize pinacolone, although constituting 57.6% of the theoretical yield. The low yield emanated from diverse sources of errors that are inherent in the experimental procedure. A feasible source of error could be an incomplete conversion of pinacol. Other possible sources of errors could be inaccurate pipetting of aqueous layer resulting in the loss of pinacolone and evaporation during the distillation process.
Sulfuric acid was used in the experiment to provide hydrogen ions (H+) for the pinacol rearrangement to occur. As -OH group is bad leaving group, its protonation results in the formation of oxonium ion (-OH2+), which is a good leaving group (Anslyn and Dougherty 675). Fundamentally, part of the reaction of pinacol rearrangement is the dehydration. Sulfuric acid dehydrates pinacol and creates tertiary carbocation, which triggers a shift in methyl group from the primary to the tertiary position.
Simple distillation was used in the separation of water and pinacolone. The simple distillation applies to the separation of two or more liquids, which have a considerable difference in boiling points (Diwekar 3). The simple distillation aimed to separate water and pinacolone from the reaction contents containing traces of pinacol and sulfuric acid. Water and pinacolone have boiling points of 100°C and 106°C respectively. As the boiling point of sulfuric acid is 290°C while that of pinacol is 172°C, these boiling points are considerably higher than that of water and pinacolone, and thus, a simple distillation procedure separates them effectively.
The distillation process stopped at 100°C because water and pinacolone have close boiling points. Raoult’s law explains why the distillation process was stopped when the temperature reached 100°C. Raoult’s principle holds that two liquids with close boiling points co-distill at the temperature that is lower than the boiling temperature of either of them (Reger, Goode, and Ball 496). The principle, therefore, implies that all water and pinacolone would have been distilled by the time the temperature reached 100°C.
Saturated sodium chloride is a liquid drying agent that has can remove a lot of water from an organic phase. As distillate has aqueous and inorganic phases, the addition of saturated sodium chloride absorbs water from the organic phase to the aqueous phase. The following chemical equation demonstrates the drying effect of saturated sodium chloride.
NaCl (s) + H2O (I) → Na+(aq) + Cl–(aq) + H2O (I)
Sodium Chloride Water Sodium ion Chloride ion water
(Pavia 716)
Since distillate has two layers, the aqueous layer has water while the organic layer has the product. Water is in the aqueous layer because it is an inorganic substance. The product is in the organic layer because pinacolone is a ketone, which is an organic substance. Comparison of the densities reveals that water is denser than the product. The density of water is 1 g/ml while the density of pinacolone is 0.801 g/ml. Therefore, aqueous phase forms the lower while the organic phase forms the upper layer in the flask.
The purpose of the anhydrous was to dry the product. Anhydrous sodium sulfate is a solid drying agent that can absorb a lot of water from another substance. Pavia states that anhydrous sodium sulfate is a hygroscopic substance that can absorb moisture from the air and become a solution (715). The following chemical equation indicates that anhydrous sodium sulfate absorbs water, which turns it into sodium and sulfate ions.
Na2SO4(s) + H2O (l) → 2Na+ (aq) + SO4-2 (aq) + H2O (l)
Sodium sulfate Water Sodium ion Sulfate ion Water
(Pavia 716)
The use of drying agents in the experiment is necessary because the organic product is mixed with water. Given that the product is an organic compound mixed with aqueous solution, saturated sodium chloride was used to remove water from the organic phase. Moreover, as the distillate comprises water and pinacolone, anhydrous sodium sulfate was used to remove water and leave pinacolone as a pure compound.
Analysis of the IR spectra of the reactant (pinacol) and the product (pinacolone) reveals that they have marked differences. As the reactant is alcohol, it exhibits IR spectrum of alcohols. The reactant has a broad O-H stretch at 3440.25 -1cm and C-H stretching frequencies at 2984.99 cm-1 and 2942.41 cm-1. In contrast, the product has C-H stretching frequencies at 2968.88 cm-1 and 2873.38 cm-1 and a unique C=O stretch at 1705.45 cm-1. Comparison of the IR spectra of the reactant and product shows that pinacol rearrangement took place. The O-H stretch is evident in the reactant because it is an alcohol while the product lacks it because it is a ketone with C=O stretch.
Moreover, NMR analysis of the reactant and the product indicates that there is an apparent chemical shift. The structure of the reactant (pinacol) comprises two tertiary hydroxyl groups and four primary methyl groups. Two protons in the hydroxyl groups and 12 protons in methyl groups depict three peaks in the NMR spectrum. In contrast, the NMR of the product illustrates chemical shifts in the spectrum. The molecular structure of pinacolone shows that it has three tertiary methyl groups, secondary carbonyl group, and secondary methyl group (Aggarwa, Kimpe, Collier, Dadoub, and Eberbach 969). The protons in these groups give rise to three types of peaks. However, the peaks are magnified because of the tertiary position of the protons and the existence of the carbonyl group in the molecular structure. Therefore, analysis of the NMR spectra confirms that there was pinacol rearrangement that resulted in the formation of pinacolone.
The 2,4-DNP test is a qualitative test that detects the presence of carbonyl groups in ketones and aldehydes. The reagent, 2,4-DNP, reacts with the carbonyl groups in solutions and forms a yellow, red, or orange precipitate (Ahluwalia and Dhingra 20). The reaction of 2,4-DNP with pinacol would not give white precipitate because it is an alcohol without carbonyl group. However, the reaction of 2,4-DNP with the product formed a yellow precipitate. The formation of a yellow precipitate confirmed that the product has a carbonyl group, and thus, it is pinacolone.
Works Cited
Aggarwa, Varinder, Norbet Kimpe, Steven Collier, Miguel Dadoub, and Wolfgang Eberbach. Science of Synthesis: Houben-Weyl Methods of Molecular Transformations: Heteroatom Analogues of Aldehydes and Ketones. New York: Georg Thieme Verlag, 2014.Print.
Ahluwalia, Vander, and Sunita Dhingra. Comprehensive Practical Organic Chemistry: Qualitative Analysis. Hyderabad: India Universities Press, 2004. Print.
Anslyn, Eric, and Dennis Dougherty. Modern Physical Organic Chemistry. Sausalito, Calif: University Science Books, 2006. Print.
Diwekar, Urmila. Batch Distillation: Simulation, Optimal Design, and Control. New York: CRC Press, 2011. Print.
Pavia, Donald. Introduction to Organic Laboratory Techniques: A Small-Scale Approach. Belmont, Calif: Thomson Brooks/Cole, 2005. Print.
Reger, Daniel, Scott Goode, and David Ball. Chemistry: Principles and Practice. Belmont: Brooks/Cole, Cengage Learning, 2010. Print.